2 Minute Medicine Rewind December 11, 2017
Contemporary Hormonal Contraception and the Risk of Breast Cancer
While it is well established that estrogen increases the risk of developing breast cancer, studies of breast cancer risk among women who use hormonal contraception have produced inconsistent results. Furthermore, few studies have investigated the risk associated with new methods, such as progestin-only, non-oral, and low-dose oral hormonal contraception. In this prospective, population-based cohort study, 1,797,932 women age 15 to 49 years from the Danish Sex Hormone Register Study (1995 to 2012) were followed up to estimate breast cancer risk among women of reproductive age using currently available hormonal contraception. The National Register of Medicinal Product Statistics provided data on filled prescriptions, while the Danish Cancer Registry provided data on primary invasive breast cancers. Researchers found that compared to women who had never used hormonal contraceptives, the relative risk of breast cancer among all current or recent users of any hormonal contraception was increased (RR 1.20, 95% CI 1.14 to 1.26, p= 0.002). The risk of breast cancer was also increased in individuals with over 10 years of use (RR 1.38, 95% CI 1.26 to 1.51) as compared to less than 1 year of use (RR 1.09, 95% CI 0.96 to 1.23). The absolute risk increase was 13 per 100,000 women overall, but in women less than 35 years, this was only 2 per 100,000 women. There were no significant differences in breast cancer risk between users of different forms of combined oral contraceptives. This study was limited by information not available in the database, such as exposure status to hormonal contraceptives before study entry. Overall, the results of this study indicate that the risk of breast cancer may be higher among women who currently or recently used contemporary hormonal contraceptives. However, this study also demonstrates a low increase in absolute risk of breast cancer among users, especially in women under 35 years old. These findings highlight the importance of balancing the risk of breast cancer against the benefits of hormonal contraceptive use, such as reduced risks of ovarian and endometrial cancer.
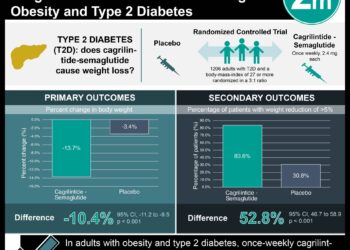
Approximately 422 million adults worldwide suffer from type 2 diabetes. Most current management approaches focus on reducing blood glucose levels and the concurrent risks of cardiovascular disease through pharmacotherapy. Prior studies have demonstrated that weight loss of at least 10 to 15 kg can lead to blood glucose normalization in patients with short-duration type 2 diabetes, however, the impact of dietary change on sustained disease remission has not been evaluated. In this randomized controlled trial, 306 non-insulin dependent type 2 diabetic patients were randomized in a 1:1 ratio to an evidence-based weight management program (Counterweight-Plus) or best-practice care by guidelines (control), with both regimens provided by routine primary care staff. The Counterweight-Plus program included a total diet replacement phase for 3 months, followed by a 2-8 week long structured food reintroduction phase, and monthly visits for long-term weight loss maintenance. Daily physical activity and sleep were monitored and exercise was encouraged, but these were not official features of the program. The co-primary outcomes were a 15 kg or more reduction in weight, and remission of diabetes to a HbA1c of less than 6.5% after at least 2 months off all diabetes medications, from baseline to 12 months. Researchers found that at 12 months, 24% of patients in the intervention group and 0% of patients in the control group had experienced a weight loss of 15 kg or more (p< 0.0001). Additionally, 46% of the intervention group and 4% of the control group achieved diabetes remission at 12 months (OR 19.7, 95% CI 7.8 to 49.8, p<0.0001). Overall, these results confirm that individuals with type 2 diabetes are more likely to enter remission through use of an evidence-based structured weight management program. Importantly, this program was dynamically tailored to the needs of each patient in the intervention group to optimize individual results. In addition, there was a dropout rate of 25% in the intervention group, possibly reflecting the difficulty of adhering to the Counterweight-Plus program. Follow-up of the study cohort will continue for at least 4 years, which should provide further insight into the long-term outcomes of such interventions.
Hemophilia B Gene Therapy with a High-Specific-Activity Factor IX Variant
Hemophilia B is an X-linked disorder characterized by recurrent bleeding episodes, resulting from deficiency or dysfunction of coagulation factor IX. The current standard of care is intravenous infusion of exogenous factor IX multiple times per week, either to prevent or respond to bleeding. However, this approach is clinically unreliable, and greatly impedes daily activities of patients. Endogenous factor IX expression after gene therapy is an appealing option. Previous studies have faltered due to insufficient factor IX expression levels, short-lived circulation, or adverse events. In this pharmaceutical-sponsored, nonrandomized study, 10 men with hemophilia B (factor IX level of ≤ 2% of normal) received a dose of 5 x 1011 vector genomes per kg of body weight of a single-stranded recombinant adeno-associated viral vector, SPK-9001, expressing a high-specific-activity factor IX variant (factor IX-R338L) to study safety and sustained factor IX activity. Zero serious adverse events and 40 adverse events occurred, 1 of which was deemed related to the study agent. This event was a transient grade 1 elevation of the alanine aminotransferase (ALT) level, which subsided following treatment with prednisone. Notably, factor IX-R338L reached steady expression in all patients by week 14, with a mean vector-derived factor IX coagulant activity of 33.7±18.5% (range 14 to 81) of normal. Moreover, the annualized bleeding rate significantly decreased following vector administration, from 11.1 to 0.4 events per year (p=0.02), and 8 of 10 patients experienced a 100% reduction in exogenous factor IX use. This study therefore shows that sustained therapeutic expression of factor IX coagulant activity can be achieved after gene transfer in patients with hemophilia B, with an acceptable patient safety profile. The long-term safety and efficacy of this approach will be determined through further follow-up of the study participants and larger trials in the future.
Critical congenital heart disease occurs in 200 per 100,000 births. In an attempt to decrease the incidence, critical congenital heart disease was added to the US Recommended Uniform Screening Panel for newborns in 2011. Subsequently, most US states enacted legislation or recommendations on pulse oximetry screening of newborns. Whether these initiatives have led to a decrease in infant death rates, however, is unknown. In this observational study, researchers analyzed US Period Linked Birth/Infant Death Data Set files from the National Center for Health Statistics (2007-2013), encompassing 26,546,503 US births, to study the impact of newborn screening on number of early infant deaths due to congenital heart disease. Between 2011 and 2013, 8 states implemented mandatory screening policies, 5 had voluntary policies, and 9 had adopted but not yet implemented policies. Researchers found that in states with mandatory screening policies, early infant deaths from critical congenital heart disease decreased by 33.4% (95% CI 10.6% to 50.3%), with an absolute decrease of 3.9 (95% CI 3.6 to 4.1) deaths per 100,000 births. Additionally, there was a relative decline of 21.4% (95% CI 6.9% to 33.7%) and an absolute decline of 3.5 deaths per 100,000 births (95% CI 3.2 to 3.8) in other or unspecified congenital cardiac deaths. Overall, the results of this study indicate the success of newborn screening policies for critical congenital heart disease in decreasing infant cardiac death. The authors recommend replication of this study in future years due to the lack of reliable information regarding actual screening observance rates and individual screening results at specific hospitals.
In patients presenting with acute kidney injury (AKI), approximately 30% of cases are caused by myeloma cast nephropathy secondary to symptomatic multiple myeloma. Standard treatment of cast nephropathy includes adequate fluid replacement, correction of precipitating factors, administration of high-dose steroids, and chemotherapy. However, the benefit of mechanically removing circulating monoclonal light chains is unclear. In this randomized controlled trial, 98 patients were randomized in a 1:1 ratio to high-cutoff hemodialysis with large-pore new-generation membranes plus standard medical care, or conventional hemodialysis plus standard medical care to compare the rate of hemodialysis independence at 3 months. Researchers found that the hemodialysis independence rate at 3 months was 41.3% in the high-cutoff hemodialysis group versus 33.3% in the conventional hemodialysis group (between-group difference 8.0%, 95% CI -12.0% to 27.9%, p=0.42). At 6 months, the rate of hemodialysis independence was 56.5% in the intervention group, compared to 35.4% in the control group (between-group difference 21.1%, 95% CI 0.9% to 41.3%, p=0.04). At 12 months, rates were 60.9% and 37.5%, respectively (between-group difference 23.4%, 95% CI 3.2% to 43.5%, p=0.02). The median progression-free survival was 35 months (95% CI 26 months to not available) in the high-cutoff hemodialysis group and 20 months (95% CI to 17 to 31 months) in the conventional hemodialysis group (HR 0.57, 95% CI 0.33 to 0.99, p=0.046). Overall, the use of high-cutoff hemodialysis versus conventional hemodialysis resulted in a significantly higher rate of kidney recovery at 6 and 12 months, although there was no significant difference at 3 months. However, the study may have been underpowered to identify an early clinically important difference. In addition, as the 6- and 12-month time-points were not primary endpoints, the significant results at these time-points must be interpreted as exploratory because of the potential for type I error inflation.
Image: PD
©2017 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.