Anti-IL-17A therapy linked to sustained improvement in plaque psoriasis: The UNCOVER trials
1. Patients with moderate-to-severe plaque psoriasis treated with ixekizumab had significantly greater improvements in psoriasis compared to those treated with placebo at the end of both 12-week induction and 60-week follow-up periods.
2. There was an increase in the rate of infections, particularly candidal in nature, in the patients exposed to ixekizumab.
Evidence Rating Level: 1 (Excellent)
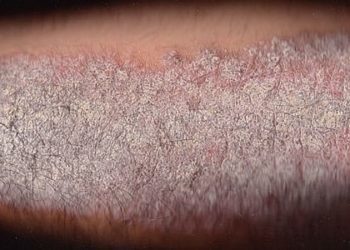
Study Rundown: Research has identified interleukin 17A (IL-17A) as an important pathogenic agent at play in chronic plaque psoriasis, a condition that has great bearing on health-related quality of life. In the UNCOVER trials 1-3, researchers investigated the efficacy of ixekizumab, a recombinant monoclonal antibody directed at IL-17A in treating plaque psoriasis. Previously published results from UNCOVER 2&3 showed that at 12 weeks, ixekizumab was superior to placebo and twice-weekly etanercept for treatment of moderate to severe psoriasis.
In this study, the authors report efficacy and combined safety results from the multicenter, randomized, double-blind, placebo-controlled UNCOVER trials across 60 weeks of follow-up. Patients were 18 years of age or older and had moderate-to-severe plaque psoriasis. At 12 weeks of follow-up, patients had a significantly better response to ixekizumab than to placebo. At 60 weeks of follow-up, patients who received continuous treatment with ixekizumab showed a sustained improvement in psoriasis symptoms. The most common adverse events during the 12-week induction period included nasopharyngitis, URIs, injection-site reactions, and headaches. Overall, oral candidiasis occurred significantly more frequently in the ixekizumab groups in both 12- and 60-week follow-up periods.
Together, the UNCOVER trials provide compelling data that targeting IL-17A is an effective treatment strategy for moderate-to-severe plaque psoriasis, and that treatment benefits can be seen over extended follow-up. However, given the chronic need for injectable treatment, further research will have to investigate the safety profile of ixekizumab over longer periods of follow-up.
Click to read the study, published today in NEJM
Relevant Reading: Comparison of ixekizumab with etan- ercept or placebo in moderate-to-severe psoriasis (UNCOVER-2 and UNCOVER-3): results from two phase 3 randomised trials
In-Depth [randomized controlled trial]: UNCOVER 1-3 involved over 100 worldwide sites and included 3866 adults with moderate-to-severe plaque psoriasis who were randomized to receive subcutaneous injections of placebo, etanercept, or ixekizumab in various permutations of treatment frequencies and dosages, over a 12-week induction phase followed by a total of 60-week follow-up period. The mean age of patients was 46, with a mean duration of psoriasis of 20 years; UNCOVER 2 & 3 specifically excluded patients who had received etanercept previously. Outcome measures included clinical measures of disease severity: the static Physician’s Global Assessment (sPGA) score and the Psoriasis Area and Severity Index (PASI).
In UNCOVER-1, the coprimary outcomes involved a 75% or greater reduction from baseline in PASI and a sPGA score of 0 (clear) or 1 (minimal psoriasis) at week 12. Ixekizumab was dosed either at 80mg every 2 or 4 weeks, and in both groups there was a significant improvement in the coprimary outcomes compared to placebo (p < 0.001 for all comparisons of ixekizumab with placebo). In the 2-wk dosing group, 81.8% had a sPGA score of 0 or 1 at 12 weeks, and 89.1% had a PASI-75 response. In UNCOVER-3, patients received ixekizumab for the 60-week follow-up period; at week 60, at least 73% had an sPGA score of 0 or 1, and at least 80% had a PASI 75 response. There were increased rates of infections, particularly candidal, in patients with ixekizumab exposure compared to placebo; however, an increase in major cardiovascular and cerebrovascular events was not noted.
Image: CC/Wiki
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.