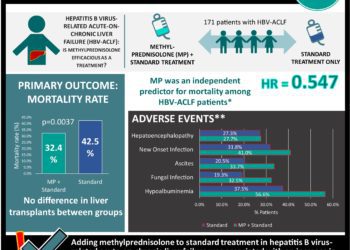
Birinapant shows efficacy in clearing hepatitis B infection [PreClinical]
1. The small molecule drug birinapant promoted elimination of hepatitis B virus (HBV) in a mouse model of chronic infection.
2. Combination therapy with birinapant and entecavir, an antiviral drug currently used to treat HBV, was more effective than either monotherapy.
Evidence Rating Level: 2 (Good)
Study Rundown: Chronic HBV infection of the liver can cause severe liver disease. While current antiviral drugs such as entecavir prevent viral replication, they do not lead to full elimination of the virus and must be taken indefinitely. The authors of this study showed in a separate report that during chronic infection, cellular inhibitor of apoptosis proteins (cIAPs) in host cells prevented the controlled death of infected hepatocytes and clearance of HBV. This subsequent study investigated the ability of birinapant, a cIAP inhibitor undergoing clinical trial evaluation for cancer treatment, to eliminate HBV during chronic infection.
In a mouse model of chronic HBV infection, birinapant treatment resulted in rapid clearance of detectable HBV genetic material from serum. Moreover, controlled cell death was observed specifically in infected hepatocytes. Combination treatment with birinapant and entecavir led to quicker clearance than treatment with either drug alone. In addition to being effective, the combination therapy did not cause significant toxicity. Overall, this study demonstrated the potential of birinapant to treat chronic HBV infection. In contrast to current antiviral drugs that target HBV itself, birinapant modulated a host cell protein involved in viral persistence. This study showed the advantage of the latter strategy in eliminating rather than merely controlling infection.
Click to read the study in PNAS
In-Depth [animal study]: This study evaluated the small molecule cIAP inhibitor birinapant for the treatment of chronic HBV infection. The C57BL/6 mouse model of chronic infection was generated by injecting the animals with plasmid that contained HBV DNA. Drug treatment was initiated one week after plasmid injection.
First, birinapant treatment was evaluated. Over the course of three weeks, animals received a weekly intraperitoneal injection of 30mg/kg birinapant or vehicle control (n=6-7 per group). Five weeks after the initial dose, there were no mice in the birinapant group with detectable serum HBV DNA. In contrast, in the group that received the vehicle control, 15% of mice (one mouse) had detectable serum HBV DNA at 12 weeks after the initial dose. Liver protein level measurements showed that birinapant decreased cIAP levels in both uninfected and infected mice. However, histology of liver tissue showed that controlled cell death promoted by birinapant occurred specifically in infected hepatocytes.
Second, birinapant and entecavir mono- and combination therapies were evaluated. During the one week of drug treatment, 30mg/kg birinapant was administered intraperitoneally at days 0 and 7 and/or 3.2mg/kg entecavir was administered orally daily (n=6 per group). Clearance of HBV DNA was significantly faster in the combination therapy group compared to birinapant alone (p<0.01), entecavir alone (p<0.001), or vehicle control (p<0.001). Specifically, no mice in the combination therapy group had detectable serum HBV DNA at two weeks after initial treatment. Notably, while both birinapant and entecavir monotherapies reduced serum HBV DNA levels below detectable levels, only birinapant also reduced serum HBV surface antigen levels below detectable levels.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.