Deep Brain Stimulation appears safe and may be effective for refractory anorexia nervosa [Physician Comment]
[tabs tab1=”2MM Rundown” tab2= “2MM Full Report”]
[tab]
Image: PD
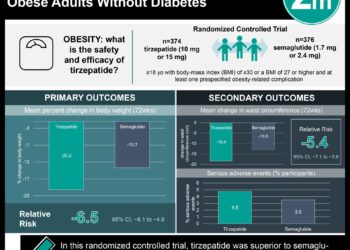
1. According to a pilot phase 1 trial, subcallosal cingulate Deep Brain Stimulation (DBS) appears to be generally safe for the treatment of refractory anorexia.
2. In some patients DBS was associated with improvements in BMI, mood, anxiety, and anorexia related symptoms.
This phase 1 clinical trial is the first to assess the use of subcallosal cingulate DBS as a treatment for chronic, severe anorexia nervosa that is not responsive to treatments. Although a small pilot trial of only 6 patients, DBS was generally safe in this study population with no episodes of stroke, infection, serious device-related complications, or death and only one adverse event (seizure during electrode programming). Improvements in BMI, mood, quality of life, and anorexia symptoms seen in some patients suggest this may be an effective treatment in otherwise refractory disease. The major limitation of this study is its small sample size, which disallows statistical analyses and limits insight into generalizability. However, these results lead the way for further research into a novel treatment for this complex and burdensome disorder that continues to affect significant mortality and morbidity.
Click to read the study in The Lancet
Click to read an accompanying editorial in The Lancet
Click here to read the clinical trial registry
[/tab]
[tab]
Image: PD
1. According to a pilot phase 1 trial, subcallosal cingulate Deep Brain Stimulation appears to be generally safe for the treatment of refractory anorexia.
2. In some patients DBS was associated with improvements in BMI, mood, anxiety, and anorexia related symptoms.
Andres M. Lozano, MD, talks to 2 Minute Medicine: Division of Neurosurgery, Toronto Western Hosptial, Toronto, Canada.
“Anorexia nervosa has the highest mortality rate of any psychiatric illness, with current management strategies largely ineffective in treating the most chronic and treatment-refractory patients. Although the physical manifestations of the illness may be the most overtly obvious, anorexia is frequently maintained by persistent psychological symptoms such as depressed mood, anxiety and affective dysregulation, which can further be linked to distinct dysfunctional neural circuits. We have conducted a trial of Deep Brain Stimulation, a neurosurgical procedure which has shown promise in other neuropsychiatric conditions, in patients with chronic anorexia nervosa. Despite evidence of safety in our small pilot trial, these promising clinical results need to be replicated in larger, placebo controlled studies, before any conclusions can be drawn regarding efficacy of DBS in anorexia nervosa”
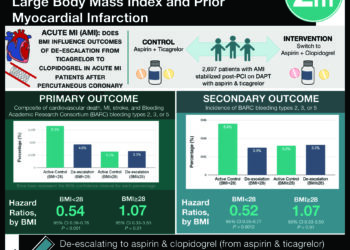
This [phase 1] trial recruited 6 patients with chronic, severe anorexia nervosa refractory to other treatments. Patients underwent neurosurgery for implantation of electrodes and pulse generators for continuous delivery of electrical stimulation to the subcallosal cingulate area and were followed for 9 months afterward. The primary outcomes evaluated were adverse events associated with surgery or stimulation, BMI, psychometrics, including anorexia symptomagology, and changes on neuroimaging.
Results found a few adverse events related to the procedure itself, including one panic attack during the surgery, one air embolus, one patient with nausea, and three patients with pain. There was one serious adverse event—a seizure that occurred two weeks after programming the device in the setting of severe metabolic abnormalities. No patients experienced infection, stroke, or death. During the nine month follow up period, four patients showed improved BMI, three reported improved mood and anorexia-related symptoms, and three reported improved quality of life.
In sum: This phase 1 clinical trial is the first to assess the use of subcallosal cingulate DBS as a treatment for chronic, severe anorexia nervosa that is not responsive to treatments. Although a small pilot trial of only 6 patients, DBS was generally safe in this study population with no episodes of stroke, infection, serious device-related complications, or death and only one adverse event. Improvements in BMI, mood, quality of life, and anorexia symptoms seen in some patients suggest this may be an effective treatment in otherwise refractory disease. The major limitation of this study is its small sample size, which disallows statistical analyses and limits insight into generalizability. However, these results lead the way for further research into a novel treatment for this complex and burdensome disorder that continues to affect significant mortality and morbidity.
Click to read the study in The Lancet
Click to read an accompanying editorial in The Lancet
Click here to read the clinical trial registry
By Maren Shapiro and Leah Hawkins
More from this author: More U.S. women using emergency contraception pill, No cost contraception reduces unintended pregnancies, Active-Duty U.S. servicewomen have higher rates of unintended pregnancy
© 2013 2minutemedicine.com. All rights reserved. No works may be reproduced without written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT. Content is produced in accordance with fair use copyrights solely and strictly for the purpose of teaching, news and criticism. No benefit, monetary or otherwise, is realized by any participants or the owner of this domain.
[/tab]
[/tabs]


![Pertussis immunity decreases after 5 DTaP doses [Researcher Comment]](https://www.2minutemedicine.com/wp-content/uploads/2013/03/9423_lores1-75x75.jpg)