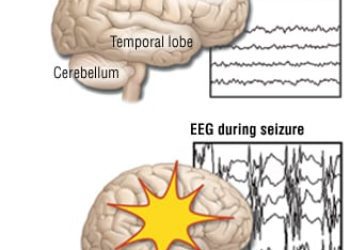
Discontinuation of antiseizure medication at discharge after acute symptomatic neonatal seizures not associated with impaired neurodevelopment
1. No significant difference in functional neurodevelopment was found at 24 months in infants who continued to take antiseizure medication (ASM) and those who discontinued ASM at time of hospital discharge following acute symptomatic neonatal seizures.
2. No significant differences were observed between cohorts in epilepsy development or gross motor function at 24 months.
Evidence Rating Level: 2 (Good)
Study Rundown: Antiseizure medications (ASMs) are often prescribed long-term for infants who suffered from acute symptomatic neonatal seizures due to concern for continued seizures and development of epilepsy; however, there is a lack of evidence to support ASM use to prevent epilepsy development, and there are significant potential adverse events from these drugs. This prospective, comparative effectiveness study compared functional neurodevelopment and risk of epilepsy at age 24 months in infants who discontinued versus maintained ASMs after discharge from neonatal seizure admission. Two-hundred seventy children (101 discontinued and 169 maintained ASMs) were evaluated at age 24 months, and there was no significant difference in the proportion of children with impaired functional neurodevelopment between groups. Furthermore, there were no significant differences between cohorts in the proportion of children who developed epilepsy or gross motor function measured at age 24 months. Overall, this study supports the notion that routine prolongation of ASMs after neonatal seizures may not be indicated in all infants. One limitation of this study, however, is that follow-up was limited to 24 months; therefore, the development of neurological impairments or epilepsy past that time point remains unknown.
Click to read the study in JAMA Neurology
Click to read an accompanying editorial in JAMA Neurology
Relevant Reading: Prophylactic phenobarbital administration after resolution of neonatal seizures: survey of current practice
In-Depth [prospective cohort]: This comparative effectiveness study enrolled 303 infants with acute symptomatic seizures from 9 neonatal seizure registry (NSR) centres between July 2015 and March 2018. Neonates with transient reversible seizure causes were excluded. The primary outcome was functional neurodevelopment at age 24 months measured using the Warner Initial Developmental Evaluation of Adaptive and Functional Skills (WIDEA-FS). Secondary outcomes included epilepsy development and functional motor impairment. Two-hundred seventy children (mean 23.8 months; 147 male) were followed up at 24 months; 101 discontinued ASMs and 169 maintained ASMs post-discharge. Median difference in WIDEA-FS scores were 4 points (2%) higher among children who discontinued ASMs (median [IQR]: 165 [150-175] versus 161 [129-174]; p= 0.09). The proportion of children with impaired functional neurodevelopment at 24 months was 28% in those who discontinued ASMs and 37% in those who maintained ASMs (OR: 0.6; 95% CI: 0.4-1.1; p= 0.11). At 24 months, 11% of children who discontinued and 14% of children who maintained ASMs developed epilepsy (OR: 0.8; 95% CI: 0.4-1.6; p= 0.49); 13% who discontinued and 19% who maintained ASMs developed functional motor impairments (OR: 0.6; 95% CI: 0.3-1.3; p= 0.18). Propensity-adjusted analyses also demonstrated no significant differences between cohorts among all outcomes.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.