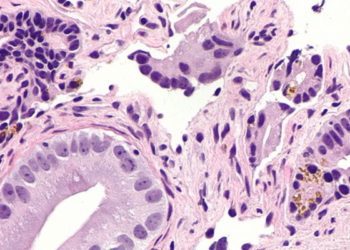
Finasteride significantly reduces incidence of prostate cancer [Classics Series]
1. Finasteride therapy was found to significantly reduce the incidence of prostate cancer compared to placebo.
2. Patients treated with finasteride were significantly more likely to have high-grade prostate cancers.
3. Patients on finasteride experienced significantly higher rates of sexual side effects, included erectile dysfunction, loss of libido, and gynecomastia
Original Date of Publication: July 17, 2003
Study Rundown: While some evidence suggests that finasteride, which inhibits the conversion of testosterone to dihydrotestosterone, reduced the risk of prostate cancer, there was no data from large, randomized, controlled trials to support this observation. In this landmark study, patients with a prostate specific antigen (PSA) of ≤3.0 ng/mL and normal rectal examination were randomized into two groups – in the first group, patients received finasteride 5 mg daily, and in the second group, patients were given placebo. Over the 7 year trial period, this study demonstrated that patients treated with finasteride experienced a significantly lower incidence of prostate cancer than those in the placebo group (RRR 24.8%; 95%CI 18.6-30.6; p<0.001). Interestingly, patients in the finasteride group had significantly higher likelihood of developing high-grade prostate cancer (37.0% vs. 22.2%, p<0.001). Patients in the treatment group had increased sexual side effects such as decreases in potency, libido, and ejaculate volumes, but also decreased urinary problems such as incontinence, frequency, and urinary tract infections. Nevertheless, in the final analysis, mortality from prostate cancer was low in both groups (5 prostate cancer-related deaths in each group).
Click to read the study in NEJM
In-Depth [randomized controlled trial] This double-blinded, randomized, controlled trial was originally published in 2003. Eligible participants were ≥55 years of age, had a normal rectal examination, and had a PSA level of ≤3.0 ng/mL. A total of 18,882 men were randomized to receive either finasteride 5 mg daily or placebo. Participants were followed for 7 years. The primary outcome measured was prevalence of prostate cancer over the course of the study. Secondary outcomes were prostate cancer mortality and tumor grade (specifically, Gleason grade ≥7) based on prostate biopsies. At the end of the study, all participants not diagnosed with prostate cancer were offered a prostate biopsy at 7 years ±90 days after randomization.
The study was terminated 15 months prior to its anticipated completion – at this time, 81.3% of participants had completed the 7 year follow-up. In the final analysis of 9,060 participants, the rate of prostate cancer diagnosis was found to be significantly lower in the finasteride group compared to placebo (RRR 24.8%; 95%CI 18.6-30.6; p<0.001). Interestingly, 48.4% of prostate cancer diagnoses were made based on end-of-study prostate biopsies, while the others were made based on cause-driven biopsies or interim procedures. Finasteride treatment was associated with higher rates of high-grade prostate cancer, defined as tumors of Gleason grade 7 or higher (37.0% vs. 22.2%, p<0.001). The use of finasteride was also associated with higher rates of various sexual side effects, including reduced ejaculate volume (60.4% vs. 47.3%), erectile dysfunction (67.4% vs. 61.5%), loss of libido (65.4% vs. 59.6%), and gynecomastia (4.5% vs. 2.8%, p<0.001 for all comparisons). Urinary urgency/frequency (12.9% vs. 15.6%), urinary retention (4.2% vs. 6.3%), and prostatitis (4.4% vs. 6.1%) rates were all significantly higher in the placebo group (p<0.001 for all comparisons). There was no significant difference in mortality from prostate cancer in the two groups (5 deaths per group).
Image: PD
© 2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.

![2 Minute Medicine: Pharma Roundup: Price Hikes, Breakthrough Approvals, Legal Showdowns, Biotech Expansion, and Europe’s Pricing Debate [May 12nd, 2025]](https://www.2minutemedicine.com/wp-content/uploads/2025/05/ChatGPT-Image-May-12-2025-at-10_22_23-AM-350x250.png)