Genetic, environmental risk assessment does not increase colon cancer screening
1. Patients who received genetic and environmental risk assessment (GERA) were not more likely to initiate colorectal cancer (CRC) screening than those who did not.
2. Among patients who received GERA, there was no significant difference in screening between those of elevated versus moderate risk for CRC.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Interactions between genetics and the environment are involved in the development of cancer, and performing risk assessments to examine susceptibility is becoming increasingly popular. However, it is unknown whether these tests could motivate recipients to improve their health behaviors. In this study, patients with average-risk for developing CRC and not adhering to the recommended screening regimen were randomized into groups that received either GERA or usual care. The GERA protocol examined the status of the methylenetetrahydrofolate reductase (MTHFR) gene and serum folate levels, which correlate with a moderate increase in risk for CRC. Although the authors predicted that GERA would encourage those at risk to subsequently follow screening guidelines, there was no significant difference in screening rates at six months post-initial assessment between the GERA and usual care groups. Within the GERA group, patients classified as having a higher risk for CRC did not pursue screening more than patients with average risk. This study was limited in that it assessed the effect of only one out of several possible sets of GERA results on one health behavior (i.e., CRC screening).
Click to read the study in the Annals of Internal Medicine
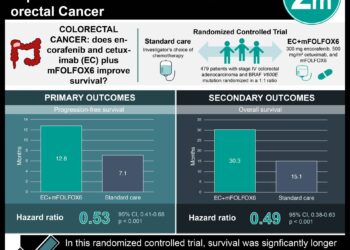
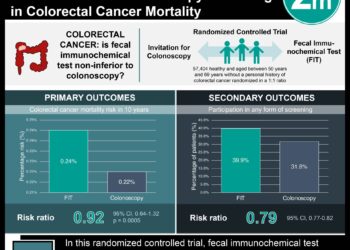
In-Depth [randomized controlled trial]: In order to be included in the study, patients needed to be between 50-79 years old, at average risk for CRC, initially non-adherent to CRC screening guidelines, off antibiotics or antifolate medications in the past six weeks, and English speaking. Out of the initial group of patients contacted, 783 completed a baseline survey and were randomized into the GERA and usual care intervention groups. Patients in the GERA group met with trained study nurses who collected blood samples for analysis, and results were subsequently given to the patients via telephone and mail. Each participant received questionnaires about CRC screening at three weeks and six months post-enrollment. After adjusting for possible confounders, the authors found that CRC screening rates were similar between groups receiving GERA and usual care (adjusted OR 0.88; 95%CI 0.64-1.22). Among patients receiving GERA, those with an elevated risk for CRC pursued screening at a lower rate than those with an average risk for CRC (adjusted OR 0.75; 95%CI 0.39-1.42).
More from this author: Financial conflicts of interest bias recommendations for influenza meds, USPSTF recommends counseling for patients at high-risk for STIs, USPSTF recommends aspirin for women at risk for preeclampsia, Marked decline in cystic fibrosis-related mortality over 10 years, ACP endorses polysomnography over portable monitors for diagnosing sleep apnea
Image: CC/Nephron
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.