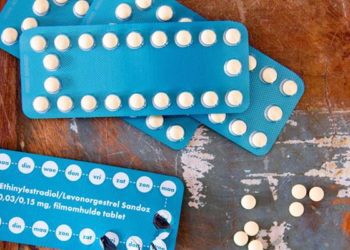
Improved contraception use with progestogen-only pill and rapid access vs emergency contraception
1. Compared to emergency contraception alone, continued use of effective contraception was 20.1% greater among women provided with a 3-month supply of progestogen-only pills and rapid access to reproductive health clinics.
2. Fewer women in the progestogen-only pill group reported using emergency contraception at 4 months post-recruitment compared to those in the emergency contraception alone group.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Emergency contraception is often used to prevent pregnancy following unprotected vaginal intercourse. Yet, most women who use oral emergency contraception remain at risk of unintended pregnancy unless an effective method of ongoing contraception is initiated. Emergency contraception is often available over the counter in many countries; however, to obtain ongoing contraception, women must speak to their general practitioner or visit a sexual and reproductive health clinic. This may take time and women might lose motivation to seek contraception, increasing the risk of unintended pregnancy. This pragmatic cluster-randomized crossover trial aimed to determine whether provision of ongoing oral contraception (progestogen-only pill) and rapid access to sexual and reproductive health clinic, combined with emergency contraception (intervention group), resulted in increased use of subsequent effective contraception compared to provision of emergency contraception alone (control group). The primary outcome for this study was self-reported use of effective contraception (hormonal or intrauterine) at 4 months. Secondary outcomes included incidence of abortion within 12 months after recruitment and economic evaluation of the intervention. Study results demonstrated that women in the intervention group were more likely to use effective contraception compared to those in the control group. In addition, more women in the intervention group reported clarity around options for ongoing contraception after discussing with a pharmacist. This randomized controlled trial was limited by a lack of generalizability as majority of the pharmacies were located in London and Edinburgh. Moreover, the follow-up period for this study was 4 months and a longer time frame could be considered to evaluate the trajectory of the study findings.
Click to read the study in The Lancet
Click to read an accompanying editorial in The Lancet
Relevant Reading: Multicenter, phase III trials on the contraceptive efficacy, tolerability and safety of a new drospirenone‐only pill
In-depth [randomized controlled trial]: Between December 19, 2017, and June 26, 2019, 29 UK pharmacies (14 in London, 12 in Edinburgh, and 3 in Tayside) and 636 participants were enrolled. The eligibility criteria included women ≥ 16 years of age, not on hormonal contraception, not taking medication that could interfere with the progestogen-only pill, and willing to provide contact information for follow-up. Women were randomly assigned to either the intervention group (n=315) or control group (n=318), with follow-up at 4 months. The mean age of participants was 22.7 years (standard deviation [SD] 5.7) in the intervention group and 22.6 years (SD 5.1) in the control group. Both groups were similar in baseline characteristics. Follow-up rates were also similar among both groups (63-65%).
The proportion of women using effective contraception (hormonal or intrauterine) was 20.1% higher (95% confidence interval [CI] 5.2-35.0, p=0.011) in the intervention group (58.4%, SD 21.6) compared to the control group (40.5%, SD 23.8). Similar findings were seen after adjusting for age, ethnicity, treatment group, study centre, previous use of effective contraception, and other factors. Compared to the control group, women in the intervention group were more likely to receive counseling on contraception from the pharmacist (76% vs. 98%); equally likely to attend the sexual and reproductive health clinic (14% vs. 17%, 95% CI -2.60% to 8.87%, p=0.284); and less likely to use subsequent emergency contraception (18% vs. 10%, 95% CI -15.38% to -1.48%, p=0.018). Overall, findings from this study suggest that provision of progestogen-only pill with emergency contraception and rapid access to care increases sustained use of effective contraception compared to provision of emergency contraception alone.
Image: PD
©2020 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.