Increasing testosterone significantly improves sexual function and depressive symptoms in older men
1. Raising serum testosterone concentrations of men 65 years of age or older, from moderately low to the mid-normal range for men 19-40 years of age, significantly increased sexual function, activity, desire, and erectile function compared to placebo.
2. The sample sizes were not large enough to draw strong conclusions as to the risks of testosterone treatment.
Evidence Rating Level: 1 (Excellent)
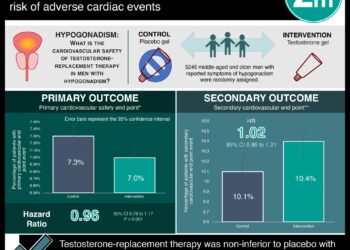
Study Rundown: As men age, serum testosterone levels decrease which leads to decreased mobility, sexual function, and energy. Prior studies on the effects of testosterone treatment in men aged 65 and older have been inconclusive. The Testosterone Trials included seven double-blind, placebo-controlled trials that were designed to understand whether supplementing testosterone would benefit older men who had low testosterone levels due to aging. Participants were 65 years or older and had a serum testosterone level that averaged less than 275 ng/dL. They received either testosterone gel or placebo gel for 1 year. Raising testosterone concentrations for 1 year significantly increased sexual activity, sexual desire and erectile function in this cohort. Testosterone had no significant effect on vitality or the percentage of men who had an increase of at least 50m in the 6-min walking distance. The rates of adverse events were similar in the two groups, however, the number of participants were too few to draw conclusions about the risks of testosterone treatment.
Click to read the study, published today in NEJM
Relevant Reading: Longitudinal Effects of Aging on Serum Total and Free Testosterone Levels in Healthy Men
In-Depth [randomized controlled trial]: The participants for this analysis were enrolled in one or more of the following trials: the Sexual Function Trial (SFT), the Physical Function Trial (PFT), or the Vitality Trial. These studies were part of the seven large multi-centre, double-blind, placebo-controlled Testosterone Trials. A total of 790 participants that were 65 years of age and had lower than 275 ng/dL serum testosterone levels were included in the analysis. They received either placebo or testosterone gel for 1 year and efficacy was assessed at 3,6,9, and 12 months. The primary outcome of the SFT was the change from baseline in the score for sexual activity on the Psychosexual Daily Questionnaire. The primary outcome of the PFT was the percentage of men who increased the distance walked in the 6-minute walk test by at least 50m. The primary outcome of the Vitality Trial was the percentage of men whose score on the FACIT–Fatigue scale increased by at least 4 points.
The analysis revealed that sexual activity, sexual desire, erectile function significantly increased in the testosterone treatment group compared to placebo (treatment effect for: sexual activity, 0.62; p<0.001; sexual desire according to the DISF-M-II, 2.93; p<0.001; erectile function, 2.64; p<0.001). There were no significant differences between the testosterone group and the placebo group in the percentage of men whose 6-min walking distance increased by at least 50m (OR, 1.42; p=0.20). However, they were more likely than those who received placebo to perceive that their walking ability had improved since the beginning of the trial (p=0.002). There was no significant difference with respect to vitality between the two groups (OR,1.23; p=0.30). There were no significant interactions of treatment with age, BMI, or race.
Image: PD
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.