Influenza vaccination in pregnancy protects both mother and infant
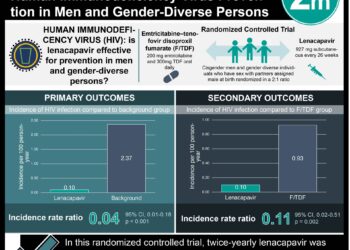
1. Seasonal influenza vaccination protected HIV-uninfected and HIV-infected pregnant women against confirmed influenza.
2. In infants from HIV-uninfected mothers, the benefits of this protection extended for up to 24 weeks post-partum.
Evidence Rating Level: 1 (Excellent)
Study Rundown: During pregnancy, the mother is in an immunocompromised state of health. As such, seasonal influenza vaccination in pregnant females is highly recommended. Prior trials have demonstrated that vaccination confers similar benefits to the infant. However, the efficacy of aggressively vaccinating pregnant patients has not been evaluated until now.
This study was comprised of two randomized, double-blinded, placebo-controlled trials from a single center in South Africa. The primary trial randomized HIV-uninfected pregnant patients to either inactivated influenza vaccination (IIV3) or placebo and the second trial used similar randomization for HIV-infected pregnant patients. Baseline characteristics were well matched for the two HIV-uninfected groups, however, there were statistical differences between the proportion of patients on antiretroviral therapy (ART) in the HIV-infected pregnant patients. Both groups of pregnant women who received vaccination had fewer cases of confirmed influenza regardless of HIV status.
Given regional and seasonal variations of influenza strains and infectivity, the results of this study may not be entirely generalized to the North American pregnant population. Since this study was powered mainly for the primary trial involving HIV-uninfected pregnant women, the results for the second trial (HIV-infected pregnant women) will also require further evaluation in a larger cohort.
Click to read the study, published today in NEJM
Relevant Reading: Influenza Vaccine Given to Pregnant Women Reduces Hospitalization Due to Influenza in Their Infants
In-Depth [randomized controlled trial]: This study encompassed two trials from a single-center in South Africa. The primary endpoint of vaccine efficacy in infants up to 24 weeks post-partum was based on the proportion of RT-PCR confirmed cases of influenza. The first trial randomized 2,116 HIV-uninfected pregnant women with well-matched baseline characteristics to receive influenza vaccination versus placebo. RT-PCR confirmed influenza occurred in 1.8% versus 3.6% (IIV3 vs. placebo, p=0.01) for mothers and 1.9% versus 3.6% (IIV3 vs. placebo, p=0.01) for infants up to 24 weeks post-partum.
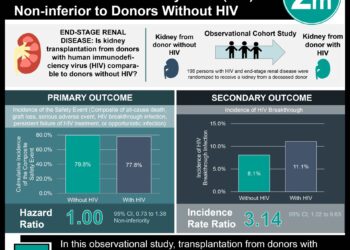
The second study randomized 194 HIV-infected pregnant women to receive influenza vaccination or placebo. Baseline characteristics that differed between the two groups included the proportion on HAART at enrollment (IIV3 vs. placebo, 22.0% vs. 40.4%). RT-PCR confirmed influenza occurred in 7.0% versus 17% (IIV3 vs. placebo, p=0.05) for mothers and 5.0% versus 6.8% (IIV3 vs. placebo, p=0.60).
Image: PD/CDC
More from this author: Brodalumab effective in treating active psoriatic arthritis, Idelalisib plus rituximab improves progression-free survival in relapsed CLL, SSRI use during pregnancy not linked to increased risk of autism,Combo antifungal therapy most effective treatment for cryptococcal meningitis
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.