Intravitreal melphalan may control retinoblastoma seeds
Image: PD
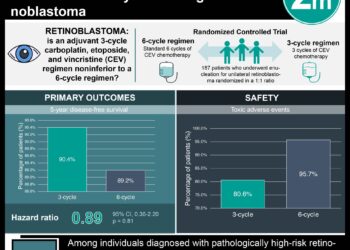
1. Monthly intravitreal injections of 20 to 30 µg/0.1 mL melphalan were administered to 11 consecutive eyes across 11 patients.
2. Administration of a mean of 5 intravitreal injections per eye led to complete regression of vitreous seeds; no recurrence was noted over an average follow-up time of 9 months.
Evidence Rating: 2 (Good)
Study Rundown: In cases of retinoblastoma, vitreous seeds of cancerous cells provide a daunting therapeutic challenge. Due to their location and distinct from the blood supply, vitreous seeds are often minimally responsive to intravenous or intra-arterial chemotherapy. This study investigated the efficacy and complications of intravitreal injections of melphalan for control of vitreous retinoblastoma seeds. Though dosing was typically continued over 6 injections, after an average of 2 injections, complete resolution of the vitreous seeds was observed with no recurrence noted within a 9-month follow-up period. This study provides preliminary evidence that intravitreal melphalan may resolve vitreous retinoblastoma seeds with only minimal complications. More studies are required to determine the stability of these results, investigate potential long-term complications, and to determine the feasibility of this treatment for subretinal seeds and solid intraretinal tumors.
Click to read the study in JAMA Ophthalmology
Relevant Reading: Intravitreal melphalan for refractory or recurrent vitreous seeding from retinoblastoma
In-Depth [retrospective cohort]: This retrospective study gauged the efficacy and complications of intravitreal melphalan injections in 11 consecutive eyes of 11 patients with retinoblastoma previously treated for their primary tumor with intravenous (5 patients) or intra-arterial (6 patients) chemotherapy. At an average age of 37 months, persistent or recurrent viable vitreous seeds were observed in all patients after primary treatment. All patients received intravitreal injections of 20 to 30 µg/0.1 mL melphalan at monthly intervals. In order to minimize the risk of extraocular seeding of cancerous tissue, melphalan injections were placed near, not directly at, the site of vitreous seeding, and triple-freeze cryotherapy was applied to the site of injection.
The study proposed that all patients should receive 6 injections, however fewer doses were provided if complete vitreous seed control was achieved or at the family’s request. A mean of 5 injections per eye were administered. Complete resolution of the vitreous seeds was observed in all 11 eyes. A mean of 2 injections were required to observe the complete response, though more injections were often administered after this point. After a mean follow-up time of 9 months, none of the 11 eyes showed signs of recurrence. There was no aspiration of vitreous fluid or tumor dissemination observed during this study. No other major complications such as vitreous infection, hemorrhage, or retinal detachment were observed. However, minor complications included 2 eyes presenting with minor focal pigment epithelial mottling and 2 eyes with extra-axial posterior lens opacity. There were no signs of retinal edema or vasculopathy.
By Michael Milligan and James Jiang
More from this author: Intranasal oxytocin may improve sociocommunicational deficits in autism
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.