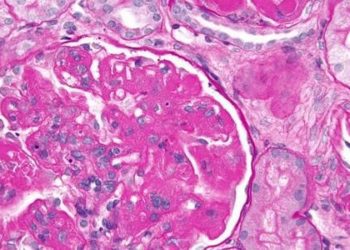
Multidrug induction therapy increases remission from lupus nephritis
1. Multi-target induction therapy for lupus nephritis with mycophenolate mofetil, tacrolimus, and steroids increased remission rates after 24 weeks compared to IV cyclophosphamide and steroids.
2. More patients in the multi-target treatment group dropped out due to adverse events.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Current induction therapy regimens for lupus nephritis (LN), a heterogeneous disease caused by systemic lupus erythematosus (SLE), have achieved low rates of complete remission. This multicenter, randomized, open-label trial sought to test if multi-target treatment (MTT) with mycophenolate mofetil, tacrolimus and steroids for recently diagnosed, biopsy-confirmed LN was superior to the current regimen consisting of IV cyclophosphamide and steroids (IVCY). Patients in the MTT group experienced significantly higher rates of complete and overall remission, and complete remission was achieved in shorter when compared to patients in the IVCY group. The rates of adverse events were similar in the two groups. More patients in the MTT group, however, stopped treatment because of adverse effects. This study was limited by the lack of a parallel group receiving mycophenolate mofetil alone, which was noted as another current induction regimen. Moreover, the study was conducted at 26 centers in China, which may limit its generalizability because of its homogeneous population. Overall, these results suggest that MTT may represent an alternative induction regimen for LN.
Click to read the study, published today in the Annals of Internal Medicine
Relevant Reading: Immunosuppressive therapies for the induction treatment of proliferative lupus nephritis: a systematic review and network metaanalysis.
In-Depth [randomized, controlled trial]: In this study, 368 subjects with recent biopsy-proven LN and confirmed SLE from 26 centers in China were randomized 1:1 to receive MTT or IVCY and evaluated at 2, 4, 8, 12, 16, 20 and 24 weeks. The primary endpoint of complete remission was significantly higher in the MTT group compared to IVCY (45.9% vs. 25.6%; difference 20.3%; 95%CI 10.0-30.6%). Patients treated with MTT experienced significantly greater rates of overall response (i.e., either complete or partial remission) (83.5% vs. 63.0%; difference 20.4%; 95%CI 10.3-30.6%) and shorter time to response compared to patients treated with IVCY (8.9 weeks vs. 13.0 weeks; difference -4.1 weeks; 95%CI -7.9 to -2.1 weeks). The rate of adverse events were not significantly different in the two groups (50.3% vs. 52.5%).
More from this author: Sofosbuvir plus ledipasvir may be effective in chronic HCV after viral relapse, Clinicians frequently unaware of the presence of central venous catheters, USPSTF recommends routinely screening asymptomatic women for chlamydia and gonorrhea, Key drugs reduce fracture risk in osteoporosis, Troponin use for suspected coronary syndrome limited in chronic kidney disease
Image: PD
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.