New antiviral drug pritelivir may reduce symptoms of HSV-2
Image: PD
1. Patients with HSV-2 treated with the helicase-primase inhibitor, pritelivir, had reduced viral shedding over 28 days when compared to patients treated with placebo.
2. Patients treated with pritelivir also experienced fewer days with symptomatic genital lesions than those treated with placebo.
3. Both viral shedding and days with genital lesions correlated with pritelivir treatment in a dose-dependent manner.
Evidence Rating Level: 1 (Excellent)
Study Rundown: The current mainstays of treatment of genital HSV infections are the DNA polymerase inhibitors valacyclovir and acyclovir – both of which require activation by viral thymidine kinase. These compounds only partially reduce transmission risk of HSV and are waning in effectiveness due to increased rates of viral resistance, particularly in immune-compromised patients. In this study, Wald et. al. tested the safety and efficacy of pritelivir, a new antiviral drug that targets the helicase-primase complex of HSV and does not require viral phosphorylation. Though the study was quite small (approx 30 participants per intervention group) it impressively demonstrated suppression of viral load in a dose dependent manner over a period of 28 days of treatment. The results also showed a dose dependent reduction in viral lesions. The study excluded immune-compromised patients, the group most likely to benefit from a novel form of treatment. Their findings, however, provide the groundwork for future studies examining the effectiveness and safety of this new class of antiviral drugs. Of note, while there were very few serious adverse effects noted in this study, the FDA has temporarily halted the clinical development of pritelivir due to unexplained dermal and hematologic findings in a toxicology study of monkeys.
Click to read the study in NEJM
Click to read an accompanying editorial in NEJM
Relevant Reading: Antiviral drug resistance and helicase-primase inhibitors of herpes simplex virus
Study Author, Dr. Anna Wald, MD MPH, talks to 2 Minute Medicine: Professor, Division of Allergy and Infectious Diseases, University of Washington
“Our industry-sponsored clinical trial shows the potential of an antiviral for hsv with an novel mechanism of action. The study design, which focused on evaluation of viral shedding, clearly demonstrated a dose-response and allows for selection of optimal dose for further studies with larger number of patients.”
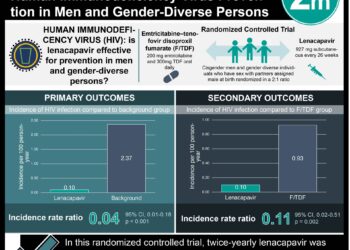
In-Depth [randomized controlled trial]: This study examined the safety and efficacy of the helicase-primase inhibitor pritelivir in adults with HSV-2 infection. The authors enrolled a total of 156 otherwise healthy adults who were seropositive for HSV-2 and had a clinical history of genital herpes. The authors assigned patients into 5 study groups and randomized them to receive one of 5 interventions over a period of 28 days: placebo, or pritelivir at a daily dose of 5mg, 25mg, 75mg, or 400mg. Patients then self-obtained daily swabs of genital skin and mucosa and provided them to the researchers for quantitative PCR detection of HSV. Patients also kept daily logs of signs and symptoms of their HSV infections. 147 of the 156 patients completed the study. The authors found viral shedding to be present on 16.6%, 18.2%, 9.3%, 2.1% and 5.3% of days in the placebo, pritelivir 5mg, 25mg, 75mg and 400mg groups respectively. The relative risk of viral shedding with pritelivir at the 75mg dose when compared to placebo was 0.13 (95% CI, 0.04 to 0.38). The relative risk of days with genital lesions in patients receiving 75mg of pritelivir versus placebo was 0.13 (95% CI, 0.02 to 0.70). The most common adverse events were headache and nausea – both of which were highest among the group receiving pritelivir at 400mg.
By Akira Shishido, MD and Xu Gao
© 2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.