Obeticholic acid associated with improved PBC biomarker trends compared to ursodiol: The POISE trial
1. Obeticholic acid therapy in patients with primary biliary cholangitis (PBC) associated with significantly greater decreases in alkaline phosphatase (ALP) and total bilirubin over 12 month period compared to placebo.
2. Higher rates of pruritis and subsequent discontinuation in patients treated with obeticholic acid compared to placebo.
Evidence Rating Level: 1 (Excellent)
Study Rundown: PBC is an autoimmune disorder of the intralobular bile ducts that, even with ursodiol therapy, can eventually lead to cirrhosis and liver failure. Higher levels of ALP and bilirubin levels tend to correlate with worsening disease state. While ursodiol, the only approved treatment for PBC, can decrease hepatic biomarkers and delay time to transplantation by binding to the farnesoid X receptor (FXR) to inhibit bile acid production and stimulate choleresis, ALP levels often remain elevated. In contrast, obeticholic acid has an approximately 100x greater affinity for FXR. In this 12 month double-blind, placebo controlled phase 3 clinical trial, researchers evaluated the effects of obeticholic acid compared to placebo (both in the context of ursodiol baseline treatment for all groups) on levels of biomarkers for PBC. In general, researchers found that obeticholic acid significantly reduced ALP levels, total bilirubin levels and other markers of cholestasis (i.e. CRP, TNF-alpha, and cytokeratin 18) compared to placebo.
Click to read the study, published today in NEJM
Relevant Reading: Levels of alkaline phosphatase and bilirubin are surrogate end points of outcomes of patients with primary biliary cirrhosis: An international follow-up study
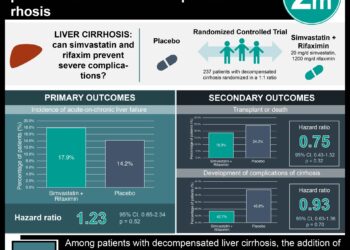
In-Depth [randomized controlled trial]: In this study, 216 patients who were 18 years or older with a diagnosis of PBC were randomly assigned to receive either 10 mg of obeticholic acid (10 mg group, n = 73) or 5 mg initially with titration up to 10 mg (5-10 mg group, n = 71) if no adverse events occurred or primary composite end point was not met, and placebo (n = 73). All groups received standard of care ursodiol therapy. The primary composite end point was an alkaline phosphatase level <1.67x the upper limit of normal with a reduction of at least 15% from baseline, and a total bilirubin less than or equal to the upper limit of normal. Overall, the primary end point was observed in significantly higher percentage of the 5-10 mg group and 10 mg group compared to the placebo group (46% and 47% vs. 10%, respective; p < 0.001 for both comparisons). Additionally, the rate of response at study interval points was significantly greater in the 5-10 mg and 10 mg group compared to placebo, as early as 2 weeks. In terms of individual components of the primary composite end point, the treatment groups also occurred at significantly higher rates of success compared to placebo. In terms of adverse effects, pruritis was more common in the obeticholic group with higher discontinuation rates, with 56% (10% discontinued) in the 5-10 mg group, 68% (1% discontinued) in the 10 mg group compared to 38% (0% discontinued) in the placebo group.
Image: CC/Wiki
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.





![Adverse pregnancy outcomes associated with thrombophilias [Classics Series]](https://www.2minutemedicine.com/wp-content/uploads/2015/07/Classics-2-Minute-Medicine-e1436017941513-75x75.png)