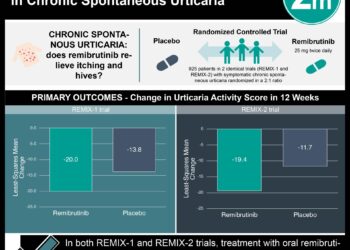
Omalizumab relieves symptoms in chronic idiopathic urticaria
[tabs tab1=”2MM Rundown” tab2= “2MM Full Report”]
[tab]
Image: PD
1. Omalizumab alleviated itchiness and reduced hives in patients with chronic idiopathic urticaria refractory to H1-antihistamine therapy.
2. Serious adverse events are more common with higher doses, to the limited extent of safety evaluations in this study.
Based on a phase-3 clinical study published today, omalizumab shows promise for alleviating symptoms in chronic idiopathic urticaria. This condition can significantly impact quality of life, especially if the itch is persistent and resistant to H1-antihistamines.
The small size of this study precludes any definitive safety conclusions. However, omalizumab has an established safety profile, and is approved for moderate-to-severe persistent asthma with a dosing regimen of 150-375 mg SC every 2-4 weeks — higher than the regimen used in this study. Clinicians might rely on this safety profile to prescribe omalizumab off-label to patients with urticaria that is severe and refractory enough to warrant use of this expensive medication.
Click to read the study in NEJM
[/tab]
[tab]
Image: PD
1. Omalizumab alleviated itchiness and reduced hives in patients with chronic idiopathic urticaria refractory to H1-antihistamine therapy.
2. Serious adverse events are more common with higher doses, to the limited extent of safety evaluations in this study.
This [randomized, phase 3] trial evaluates the use of omalizumab in treating chronic idiopathic urticaria. Participants with H1-antihistamine resistant symptoms were randomized into four groups receiving either placebo or omalizumab at doses of 75 mg, 150 mg, or 300 mg.
Groups receiving 150 mg and 300 mg of omalizumab saw significant improvements in weekly itch-severity scores, with mean changes of -8.1±6.4 (p=0.001) and -9.8±6.0 (p<0.001) from baseline, respectively. The group receiving 75 mg of omalizumab (mean change -5.9±6.5, P=0.46) did not improve significantly compared to the placebo group (mean change -5.1±5.6). The regimen was also effective in reducing urticaria with a higher proportion of patients hives-free at week 12 (10% placebo, vs. 18%, 23%, and 53% for each omalizumab dosing regimen).
Serious adverse events were most common (6%) in the group receiving 300 mg omalizumab. However, the study was too small to draw any definitive conclusions regarding safety.
In sum: Based on this phase-3 clinical study, omalizumab shows promise for alleviating symptoms in chronic idiopathic urticaria. This condition can significantly impact quality of life, especially if the itch is persistent and resistant to H1-antihistamines.
The small size of this study precludes any definitive safety conclusions. However, omalizumab has an established safety profile, and is approved for moderate-to-severe persistent asthma with a dosing regimen of 150-375 mg SC every 2-4 weeks — higher than the regimen used in this study. Clinicians might rely on this safety profile to prescribe omalizumab off-label to patients with urticaria that is severe and refractory enough to warrant use of this expensive medication.
Click to read the study in NEJM
By Joe Liu and Mitalee Patil
More from this author: New device reduces acid exposure in gastroesophageal reflux disease, Neurostimulation beneficial in Parkinson’s disease with early motor complications, Rivaroxaban has no net clinical benefit over enoxaparin for thromboprophylaxis in acutely ill patients, Long term functional outcomes similar with surgery, radiotherapy in prostate cancer patients
© 2013 2minutemedicine.com. All rights reserved. No works may be reproduced without written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT. Content is produced in accordance with fair use copyrights solely and strictly for the purpose of teaching, news and criticism. No benefit, monetary or otherwise, is realized by any participants or the owner of this domain.
[/tab]
[/tabs]

![2 Minute Medicine: Pharma Roundup: Price Hikes, Breakthrough Approvals, Legal Showdowns, Biotech Expansion, and Europe’s Pricing Debate [May 12nd, 2025]](https://www.2minutemedicine.com/wp-content/uploads/2025/05/ChatGPT-Image-May-12-2025-at-10_22_23-AM-350x250.png)