Oral RSV entry inhibitor effective at reducing viral load and clinical symptoms
1. Adults treated with the drug GS-5806 had lower respiratory syncytial virus (RSV) viral loads after 12 days when compared to adults treated with placebo.
2. No significant difference in side effects between the treatment groups was observed.
Evidence Rating Level: 1 (Excellent)
Study Rundown: RSV is responsible for causing respiratory tract infections in people of all ages, but is also the most common cause of lower respiratory tract infections in children under a year of age. Many infants are hospitalized each year for wheezing and difficulty breathing caused by RSV infection. Treatment generally involves relieving symptoms rather than targeting the virus itself.
GS-5806 is an experimental drug that interferes with RSV’s ability to infect host cells. In this study, healthy adults were experimentally exposed to RSV and then treated with either GS-5806 or placebo at different doses to determine whether the drug had any effect on viral load or symptoms. Treatment with GS-5806 was associated with lower viral load and reduced symptoms. Adverse events associated with treatment included some laboratory abnormalities which were mostly considered mild.
This was a small study with only 87 subjects in total receiving the treatment drug. While GS-5806 may perform well in a controlled laboratory setting where only one strain of virus is used, this success may not translate into efficacy in the field. Furthermore, results derived in adults may not apply to children. Safety and efficacy in children with natural infections will have to be determined in future studies. When considering any new therapy, its benefits must always be weighed against its potential harms. Bearing in mind that RSV infection typically resolves on its own, GS-5806 will have to prove itself either extremely safe or extremely efficacious to warrant wide-spread use. More likely, it will find a niche for itself in treatment of more severe cases of RSV infection.
Click to read the study, published today in NEJM
Click to read the accompanying editorial in NEJM
Relevant Reading: Global burden of acute lower respiratory infections due to respiratory syncytial virus in young children: a systematic review and meta-analysis
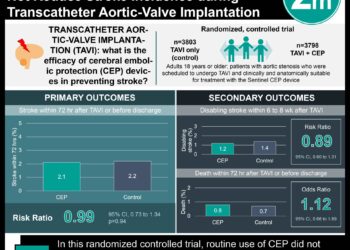
In-Depth [randomized controlled trial]: In this study, 140 adults were experimentally inoculated with RSV. Subjects were evaluated for active infection 5 days after inoculation and were then randomized to either GS-5806 or placebo regardless of infection status. Importantly, efficacy data was derived only from those who demonstrated active infection. The primary end point was the area under the curve (AUC) for the viral load, determined by quantitative RT-PCR assay. Secondary end points were the total weight of mucus production and the change in symptom scores as rated by the participants.
Groups 1-4 were randomly assigned to either placebo or 50mg of GS-5806 on day one, followed by four days of 25mg daily. Participants treated with GS-5806 had decreased viral load after 5 days of treatment as compared to those who received placebo, difference of 531.0 log10 PFUe x hours per milliliter (95% Confidence Interval, 365.3 to 696.8; P<0.001). Groups 5-7 received placebo or varying doses of GS-5806. All participants in groups 5-7 that received GS-5806, regardless of dosage, had decreased viral loads, mucous weight, and symptoms as compared to those treated with placebo.
The only adverse event the authors deemed to be related to the study drug was a grade 1 decrease in pulmonary function. This was observed in 4 of 87 (4.6%) participants in the GS-5806 group and 2 of 53 (3.8%) participants in the placebo group. 14.9% of participants treated with GS-5806 had elevated levels of alanine aminotransferase as compared to 9.4% in the placebo groups. Elevated levels of aspartate aminotransferase were less frequent with GS-5806 than with placebo (5.7% vs. 15.1%). In addition, more participants who received GS-5806 had low neutrophil counts, 11.5%, as compared to placebo, 5.7%.
Image: PD
More from this author: Early risk factor for progression of cystic fibrosis identified, Gut microbes implicated in stroke and heart attacks: new dietary link, New leukemia mutation offers therapeutic targets, Childhood ADHD associated with increased risk of suicide, A marker of aggressive liver cancer and potential therapeutic target identified
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.