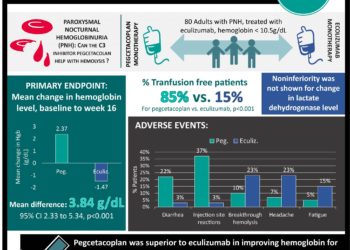
Pegcetacoplan improved hemoglobin levels in eculizumab-treated paroxysmal nocturnal hemoglobinuria patients
1. Pegcetacoplan was shown to reduce the change in hemoglobin level for the treatment of paroxysmal nocturnal hemoglobinuria compared to eculizumab.
2. Patients treated with pegcetacoplan experienced fewer transfusions and less fatigue.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Paroxysmal nocturnal hemoglobinuria (PNH) is a hematological disorder that results from aberrant hematopoietic clones that result in complement-mediated extra- and intravascular hemolysis and thrombosis. C5 inhibitors such as eculizumab help control intravascular hemolysis and are one of the standard treatments for PNH. However, extravascular hemolysis still occurs and is not targeted by C5 inhibition. Pegcetacoplan, a C3 inhibitor, is potentially effective for both extravascular and intravascular hemolysis in PNH. As such, this study compared pegcetacoplan to eculizumab in eculizumab-treated PNH patients. The study determined pegcetacoplan monotherapy had less hemoglobin loss and more transfusion-free patients. The study was limited by a small patient population. Nonetheless, this trial demonstrated that pegcetacoplan was superior to eculizumab in controlling hemoglobin loss and improved in key patient outcomes with a favorable adverse event profile.
Click here to read the study in the NEJM
In-Depth [randomized controlled trial]: This was a multicenter, randomized, open-label trial of 80 adult patients with PNH. The inclusion criteria were patients treated with eculizumab therapy and hemoglobin levels lower than 10.5 g per deciliter. Patients not treated with eculizumab for at least three months prior to the study were excluded from the study. Patients were randomized in a 1:1 ratio to either pegcetacoplan monotherapy or eculizumab monotherapy, respectively. The primary endpoint was hemoglobin level change from baseline to week 16. The mean change was 2.37 g per deciliter in the pegcetacoplan group compared to -1.47 g per deciliter in the eculizumab group (mean difference, 3.84 g per deciliter; 95% confidence interval [CI], 2.33 to 5.34; P < 0.001). More patients in pegcetacoplan group (35 out of 41 patients, 85%) were transfusion-free compared to the eculizumab group (6 out of 39 patients, 15%) (P < 0.001). Furthermore, the change in lactate dehydrogenase level from baseline did not achieve noninferiority between pegcetacoplan monotherapy (-15±43 U per liter) and eculizumab monotherapy (-10±71 U per liter). The incidence of serious adverse events was similar between both groups. The most common adverse events in the pegcetacoplan group were injection site reactions and diarrhea; however, there were no reported cases of meningitis. Overall, pegcetacoplan was superior to eculizumab in key hematological parameters and clinical outcomes with a side effect profile favoring pegcetacoplan.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.