Posaconazole is non-inferior to voriconazole for primary treatment of invasive aspergillosis
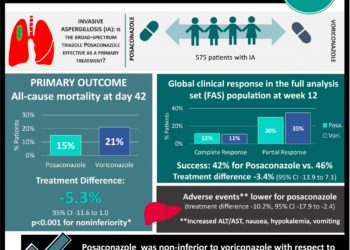
1. Posaconazole was non-inferior to voriconazole with respect to all-cause mortality at day 42 (15% vs. 21%, respectively).
2. Incidence of treatment-related adverse events was lower for posaconazole than voriconazole.
Evidence Rating Level: 2 (Good)
Study Rundown: Invasive aspergillosis (IA) is a life-threatening infection that commonly occurs in patients with immunosuppression. Currently, voriconazole serves as the primary treatment for this condition but there remain concerns regarding its acute and long-term toxicities. Posaconazole, a broad-spectrum triazole, may be used for salvage treatment of IA, but investigations of its efficacy and safety from clinical trials is lacking. This multicenter, randomized controlled trial aimed to assess the non-inferiority of posaconazole to voriconazole for the primary treatment of invasive aspergillosis. Primary endpoint for this study was cumulative all-cause mortality until day 42 in the intention-to-treat (ITT) population, while secondary endpoints included all-cause mortality until day 84 (ITT), all-cause mortality until day 42 and day 84 in the full-analysis-set (FAS) population, and global clinical response. According to the study, all-cause mortality at day 42 was non-inferior between the posaconazole and voriconazole group. In addition, posaconzaole was well-tolerated, and participants reported fewer treatment-related adverse events. This study was limited by a lack of longitudinal follow-up as outcomes were only reported over a three-month period. Nonetheless, it provides valuable insight regarding the use of posaconazole as a first-line treatment for patients with invasive aspergillosis.
Click to read the study in The Lancet
Relevant Reading: Comparison of Antifungal Prophylaxis Drugs in Patients With Hematological Disease or Undergoing Hematopoietic Stem Cell Transplantation
In-depth [randomized controlled trial]: Between Oct 25, 2013, and Sept 10, 2019, 653 patients were assessed for eligibility across 91 study sites in 26 countries. Included were those aged 13 years and older, weighing ≥ 40 kg, and meeting the criteria for proven, probable, and possible fungal disease. Patients with chronic aspergillosis (> 1-month duration), relapsed or recurrent disease, and significant liver dysfunction were excluded. Altogether, 575 patients (288 in the posaconazole group and 287 in the voriconazole group) were included in the analysis. Among enrolled patients, the median age was 57 years (interquartile range [IQR] 41-66) and 60% were male. Risk factors for IA included prolonged neutropenia (64%), treatment with T-cell immunosuppressant drugs (41%) or corticosteroids (32%), and allogenic hematopoietic stem cell transplantation (22%).
The primary endpoint concerning all-cause mortality at day 42 (intention-to-treat population) was 15% (44 of 288) in the posaconazole group and 21% (59 of 287) in the voriconazole group (treatment difference -5.3%, 95% confidence interval [CI] -11.6 to 1.0, p<0.0001). At day 84, a similar pattern was observed between both groups (28% for posaconazole vs. 31% for voriconazole, treatment difference -2.5%, 95% CI -9.9 to 4.9). The secondary endpoint of all-cause mortality (full-analysis-set population – ITT patients with proven or probable IA) from baseline to day 42 showed non-inferiority for posaconazole (19%) compared to voriconazole (19%, treatment difference 0.3%, 95% CI -8.2 to 8.8). Moreover, global clinical response in the FAS population was similar for both treatment groups at 6 weeks and 12 weeks from baseline. The incidence of treatment-related adverse event rates in the ITT population was also lower for posaconazole (30%) than voriconazole (40%, treatment difference -10.2%, 95% CI -17.9 to -2.4). Overall, findings from this study support the use of posaconazole as a first-line treatment for invasive aspergillosis given its high tolerability and minimal treatment-related side effects.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.


![Adverse pregnancy outcomes associated with thrombophilias [Classics Series]](https://www.2minutemedicine.com/wp-content/uploads/2015/07/Classics-2-Minute-Medicine-e1436017941513-350x250.png)
![New drug for Chagas disease shows antitrypanosomal activity [CHAGASAZOL trial]](https://www.2minutemedicine.com/wp-content/uploads/2014/05/t-cruzi-350x250.jpg)





