Quick Take: Veverimer versus placebo in patients with metabolic acidosis associated with chronic kidney disease
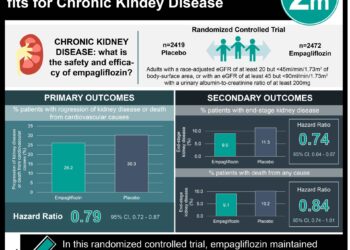
Chronic metabolic acidosis is often seen in patients with chronic kidney disease (CKD), and results from an inability to excrete metabolically produced acid. This, in turn, is associated with bone resorption, muscle catabolism, and progression of kidney disease. Veverimer is a non-absorbable medication that removes hydrochloric acid from the gut lumen in order to combat acidosis. In this multicenter, randomized controlled trial, 217 patients age 18 to 85 years with non-dialysis dependent CKD (estimated glomerular filtration rate between 20 and 40 mL/minute per 1.73 m2) with metabolic acidosis (serum bicarbonate between 12 and 20 mmol/L) were randomly assigned to receive either veverimer or placebo for 12 weeks while adhering to their regular diet to assess the efficacy and safety of veverimer as a treatment for metabolic acidosis in this patient population. Patients were followed up for the composite primary end point, which was the difference in the proportion of patients achieving either a 4 mmol/L increase in serum bicarbonate concentration from baseline or a serum bicarbonate level in the normal range of 22-29 mmol/L. Researchers found that there was a significant increase in the proportion of patients achieving the composite primary endpoint in the veverimer group, as compared to placebo (59% vs. 22%, difference 37%, 95% CI 23% to 49%, p<0.0001). The most common treatment-related adverse events observed in the interventional group were gastrointestinal, including diarrhea, flatulence, nausea, and constipation; these events were typically mild to moderate in severity. This study therefore shows that veverimer is a safe and effective therapy for metabolic acidosis in patients with CKD. Further studies are needed in investigating the long-term effect and safety of this medication.
Click to read the study in Lancet
Image: PD
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.