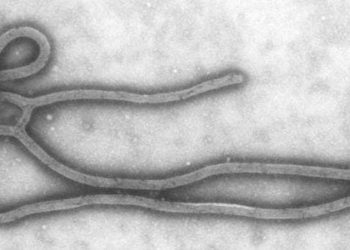
Recombinant vesicular stomatitis virus Ebola vaccine demonstrates immunogenicity
1. Zaire ebolavirus-glycoprotein-specific antibody responses were detected in all participants receiving vaccination doses from 300,000 to 50 million plaque-forming units (PFU).
2. Fever was reported in 13-30% of participants receiving the recombinant vesicular stomatitis virus-vectored Zaire ebolavirus (rVSV-ZEBOV) vaccination.
Evidence Rating Level: 2 (Good)
Study Rundown: In August 2014, the WHO declared the Ebola outbreak to be a public health emergency. Subsequently, the Public Health Agency of Canada donated 800 vials of replication-competent rVSV-ZEBOV trial vaccine to the WHO, which was found to have demonstrated efficacy in nonhuman primates. In this study, researchers investigated the 6-month safety, side effect profile, reactogenicity and immunogenicity of the rVSV-ZEBOV vaccine through three dose-escalation trials and one randomized, double-blind controlled phase 1 trial. With regards to safety, minimal adverse events were reported. The authors note the vaccine’s side effect profile included fever in approximately 13-30% of participants. Levels of rVSV RNA were transiently detected in almost all participants’ serum samples. Most notably, the rVSV-ZEBOV vaccine generated glycoporiten-binding antibodies in all participants at any dose, with persistence through 180 days.
Click to read the study in NEJM
Relevant Reading: Recombinant vesicular stomatitis virus-based vaccines against Ebola and Marburg virus infections
In-Depth [randomized controlled trial]: In this trial, researchers ran three open-label dose-escalation phase 1 trials, and one randomized, double-blind controlled phase 1 trial to determine the safety, reactogenicity, immunogenicity and side-effect profile of the rVSV-ZEBOV vaccine with a total of 158 participants. The three open-label dose-escalation trials took place in Lambarene, Hamburg and Kilifi from November 2014 to January 2015. The randomized controlled trial was based in Geneva from November 2014 to December 2014. In the Lambarene trial, participants received the vaccine at 300,000 PFU (N=20) up to 3 million PFU (N=19). In the Kilifi and Hamburg trials, participants received the vaccine at 3 million PFU (Kilifi N=20; Hamburg N=10) and up to 20 million PFU (Kilifi N=20, Hamburg, N=10). In the Geneva trial, participants received the vaccine at 10 million PFU (N=35) up to 50 million PFU (N=16) and received volume-matched saline placebo (N=8).
Fever was observed in approximately 13-30% of vaccines, with objective fever being recorded for 25% (5/20) of Hamburg participants, 30% (12/40) of Kilifi participants, 13% (5/39) of Lambarene participants and 21% (13/51) of Geneva participants. Vaccine viremia was identified in 40% (8/20) and 95% (18/19) in the Lambarene group for 300,000 and 3 million PFU, respectively, and 90% (46/51) in the Geneva group. Overall, for participants receiving 3 million PFU or more, at least 95% (123/130) demonstrated positive RT-PCR for rVSV within 3 days of vaccination. With the exception of 11 plasma specimens, all samples were negative by day 8 and no infectious virus was detected from any blood specimen tested. Across the three trials, saliva and urine samples did not yield viral RNA. ZEBOV-glycoprotein-specific antibody responses were detected in all vaccines at similar mean titer levels within four weeks after immunization. While the participants receiving 300,000 PFU (lowest dose) in the Lambarene trial demonstrated immunogenicity, a low response rate was more frequent compared to those receiving higher doses. In the Geneva trial, antibody titers more than doubled at the 28-day mark in 33% (41/126) participants and reached significantly higher geometric mean titers (GMT = 1634 up from 1064) at 10 million PFU dosing at 180 days in Geneva (Geometric mean ratio of 180 days from 28 days titer=1.59, 95% [CI] = 1.21-2.09, p=0.004).
Image: PD
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.