Small-molecule inhibitor may enhance recovery after brain hemorrhage [PreClinical]
1. Mice deficient in hypoxia-inducible factor prolyl hydroxylase domain (HIF-PHD) enzymes exhibited improved behavioral performance after induction of intracerebral hemorrhage (ICH).
2. Adaptaquin (AQ), a HIF-PHD inhibitor, lowered neuronal death and the decline of behavioral functions in multiple rodent models of ICH.
Evidence Rating Level: 2 (Good)
Study Rundown: ICH, a subtype of stroke that can be caused by conditions including traumatic brain injury and hypertension, can lead to rapid neurological damage via mechanisms such as iron accumulation. Iron chelators have been tested for ICH treatment, but they can disrupt normal physiological functions dependent on iron. In this study, the authors sought to specifically inhibit HIF-PHDs, metalloenzymes that are targeted by the more general iron chelators.
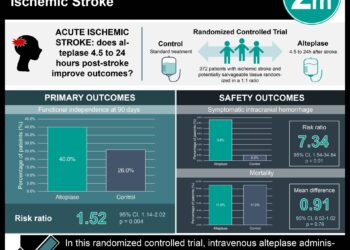
Mice with genetically reduced expression of the three HIF-PHD isoforms in the brain were subjected to injury resulting in ICH. As compared to the control cohort, these PHD-deficient mice exhibited enhanced somatosensory function several days after injury, measured by the corner task and the tape removal task. In a mouse model of ICH, inhibition of HIF-PHDs by the small molecule AQ resulted in similar improvements in behavioral performance. AQ also reduced the number of degenerating neurons in the regions surrounding the brain bleed as compared to the control vehicle-treated group. In a second rodent model of ICH where motor impairments were more long-term, rats treated with AQ performed better than those that received the vehicle control in a pellet reaching task, at both early and late stages after injury.
While additional studies to establish the overall safety of AQ are necessary, this work demonstrates the potential of using such an inhibitor to target neurological damage caused by brain hemorrhage. Incidentally, drugs similar to AQ are currently being tested in patients with neurodegenerative diseases such as Alzheimer’s disease and Huntington’s disease.
Click to read the study in Science Translational Medicine
Relevant Reading: Deferoxamine Treatment for Intracerebral Hemorrhage in Aged Rats – Therapeutic Time Window and Optimal Duration
In-Depth [animal study]: Male C57BL/6-FVB mixed mice were genetically bred to have reduced expression of PHD1, PHD2, and PHD3 in the brain striatum. ICH injury was induced in these mice by collagenase injection into the right striatum, which caused disruption of the blood vessel basal lamina, into the right striatum. Mice were evaluated for behavioral test performance 1, 3, and 7 days post-ICH. PHD-deficient animals (n=14) performed significantly better in the corner task, where mice were placed in a corner so that they reared up and turned around to face the open end. Those with reduced PHD expression showed less preference in using their ipsilateral limbs during turning 3 and 7 days after ICH (p<0.01). Likewise, PHD-deficient mice also exhibited significantly quicker response times to remove adhesive tape from their contralateral paw (p<0.01) compared to the control group 1 and 3 days post-ICH.
When normal C57BL/6 mice (n=14) were treated with AQ (30 mg/kg delivered intraperitoneally) once a day for 7 days beginning 2 hours after collagenase injection, performance in the corner task and tape removal task was significantly enhanced (p<0.01). Histology of brain sections using Fluor-Jade B staining indicated that AQ decreased the number of apoptotic/necrotic neurons in and near the brain bleed relative to the vehicle (n=7-8, p<0.001). In a second model of ICH, Sprague Dawley rats were injected with autologous blood in the striatum and treated with the same daily AQ treatment regimen. They were evaluated on their ability to reach for a food pellet behind a narrow vertical slot. AQ administration led to a significantly improved ability to reach using the contralateral limb at early (9-11 days, p=0.011) and late (25-28 days, p=0.005) time points post-ICH compared to vehicle treatment (n=14-15).
Image: PD
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.