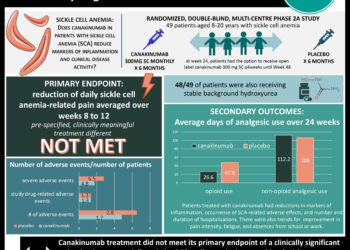
Stem cell transplant may be effective in sickle cell disease
1. 83% of sickle cell patients receiving a nonmyeloablative, hematopoetic stem cell transplant demonstrated full donor-type hemoglobin one-year post transplantation.
2. No patient has developed graft-versus-host disease thus far.
Evidence Rating Level: 2 (Good)
Study Rundown: Sickle cell disease is caused by a mutation in the beta-globin protein and results in hemolysis and end-organ damage due to vascular occlusion. While sickle cell disease can be treated with hydroxyurea and blood transfusions, the only cure is stem cell transplantation. This study utilized a nonmyeloablative conditioning regimen (alemtuzemab and whole body radiation) before conducting allogeneic stem cell transplant from an HLA-matched relative in an effort to maximize the benefit of transplantation while avoiding complications such as graft-versus-host disease. This study included 30 adults with severe sickle cell disease or B-thalassemia with an endpoint of full donor-type hemoglobin cells at 1 year for sickle cell patients and transfusion independence for B-thalassemia patients. Strengths of this study include the relatively large number of patients and the wide age range of included individuals. However, results are limited by the relatively short study follow up time. Ultimately, this study introduces a very promising potential therapy for sickle cell disease, but before it can be more widely used a larger study with longer follow up time will be needed.
Click to read the study, published today in JAMA
Click to read an accompanying editorial in JAMA
Relevant Reading: Allogeneic hematopoietic stem-cell transplantation for sickle cell disease
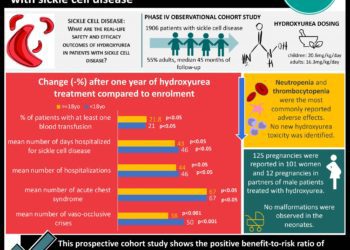
In-Depth [prospective cohort]: This study followed 30 individuals with sickle cell disease and beta-thalassemia for a median of 3.4 years after HLA-matched relative donor nonmyeloablative allogeneic stem cell transplant. 83% of patients achieved donor hemoglobin 1 year after transplant and 29 out of 30 were alive at the end of the study. At the most recent follow up time, hemoglobin levels were increased (81 g/ L versus 127 g/L in women, 89 g/L versus 137 g/L in men; p<0.001) and markers of hemolysis, such as reticulocyte count (182 x 109 cells/L versus 85 x 109 cells/L; p<0.001), total bilirubin (2.7 mg/dL versus 0.9 mg/dL; p<0.001), and lactate dehyrogenase (324 U/L versus 202 U/L; p<0.001) were decreased compared to pre-transplantation levels. The median duration of immunosuppressive therapy was 2.1 years (range 1.0-8.4) and cessation of therapy was possible in 15 out of 30 patients. No patients developed graft-versus-host disease during the study.
More from this author: Ambulances equipped for thrombolysis allow for more rapid stroke treatment, Physical therapy for hip osteoarthritis may not lead to significant benefits, Smaller changes in kidney filtration may predict end stage renal disease progression, Bariatric weight loss surgery linked with remission in diabetics, No link between TNF-a inhibitor use and cancer in inflammatory bowel disease
Image: CC/Wiki/Uthman
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.