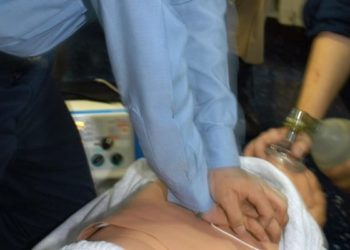
Trans-nasal cooling during cardiac arrest does not improve survival with good neurologic outcome
1. In this randomized controlled trial, intra-cardiac arrest, trans-nasal evaporative cooling did not improve survival with good neurologic outcome at 90 days compared to standard systemic cooling in the ICU.
2. The time to core target temperature was shorter in the intra-nasal cooling group.
Evidence Rating Level: 1 (Excellent)
Study Rundown: There is increasing evidence that therapeutic hypothermia may increase survival with good neurologic outcome after out-of-hospital cardiac arrest. Trans-nasal evaporative cooling is a technique that can be induced intra-arrest, can be done en-route to the hospital, and leads to continuous cooling of the brain. In this randomized controlled trial, intra-cardiac arrest, trans-nasal evaporative cooling did not improve survival with good neurologic outcome at 90 days compared with standard systemic cooling in the ICU. Despite no difference in survival with good neurologic outcome, the time to core target temperature was shorter in the intra-nasal cooling group. Minor nosebleed was the most common adverse event with intra-nasal cooling.
As the time to core temperature was shortened in the intervention group, it is unclear why this intervention did not improve survival, though the study has several limitations. Prehospital and hospital personnel were not blinded to treatment. Also, the study period was long and many eligible patients with cardiac arrest were not included in the trial. Lastly, the study may have been underpowered to detect a clinically important difference. A larger trial might likely be needed to further study the effectiveness of this intervention.
Click to read the study published today in JAMA
Relevant Reading: Design of the PRINCESS trial: pre-hospital resuscitation intra-nasal cooling effectiveness survival study (PRINCESS).
In-Depth [randomized controlled trial]: The PRINCESS trial was a randomized clinical trial that was conducted in 11 emergency medical service (EMS) systems in 7 European countries. The inclusion criterion was bystander-witnessed cardiac arrest in patients at least 18 years of age from July 2010 to January 2018. This study included 677 patients with bystander-witnessed out-of-hospital cardiac arrests (337 in the intervention and 334 in the control group). The intervention group received oxygen and perfluorohexane via nasal canula en-route to the hospital during cardiac arrest, and the control group received care as usual upon arriving to the hospital. Data on survival or functional recovery were collected by means of a structured interview over the phone or person-to-person using the Pittsburgh Cerebral Performance category (CPC) scale. The primary outcome was survival with good neurologic outcome 90 days after arrest, defined as CPC 1 or 2. The secondary outcomes were overall survival rate at 90 days and cooling efficacy, measured as time from collapse to core temperature less than 34°C. At 90 days, the number of patients who survived with good neurologic function (CPC 1-2) at 90 days was 56 of 337 (16.6%) in the intervention group vs 45 of 334 (13.5%) in the control group (difference 3.1%; CI95 −2.3 to 8.5%). For secondary outcomes, time to target core temperature was 105 minutes in the intervention group vs 182 minutes in the control group (p < .001).
Image: PD
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.