#VisualAbstract: A Randomized Trial of Prenatal n−3 Fatty Acid Supplementation and Preterm Delivery
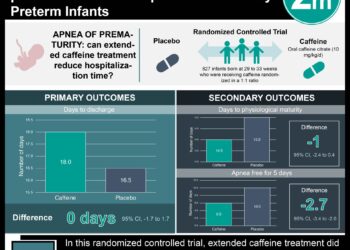
1. A multicenter randomized control trial concluded that n-3 polyunsaturated fatty acid supplementation in pregnancy did not change the risk of preterm delivery or the risk of post-term induction or cesarean section.
2. The safety profile of n-3 polyunsaturated fatty acid supplementation in pregnancy was similar to control.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Preterm delivery, delivery before 37 weeks of gestation, is associated with increased childhood morbidity and mortality. N-3 long-chain polyunsaturated fatty acids are known to play a role in modulating proinflammatory prostaglandins such as PGE2 and PGF2a that are associated with cervical ripening and uterine contractility. In this multicenter, double-blind randomized control trial, researchers assessed whether supplementation of n-3 long chain fatty acids between 14 and 34 weeks of gestation would lead to a lower rate of preterm delivery and lower neonatal complications after delivery. Participants in this study were randomized to take supplements of n-3 long chain polyunsaturated fatty acids or a control supplement largely consisting of isocaloric vegetable oil. Overall, researchers noted no difference in preterm delivery rates between groups. There also was no noted difference in the rate of post-term induction of labor or the rate of post-term cesarean section.
Click to read the study in NEJM
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc