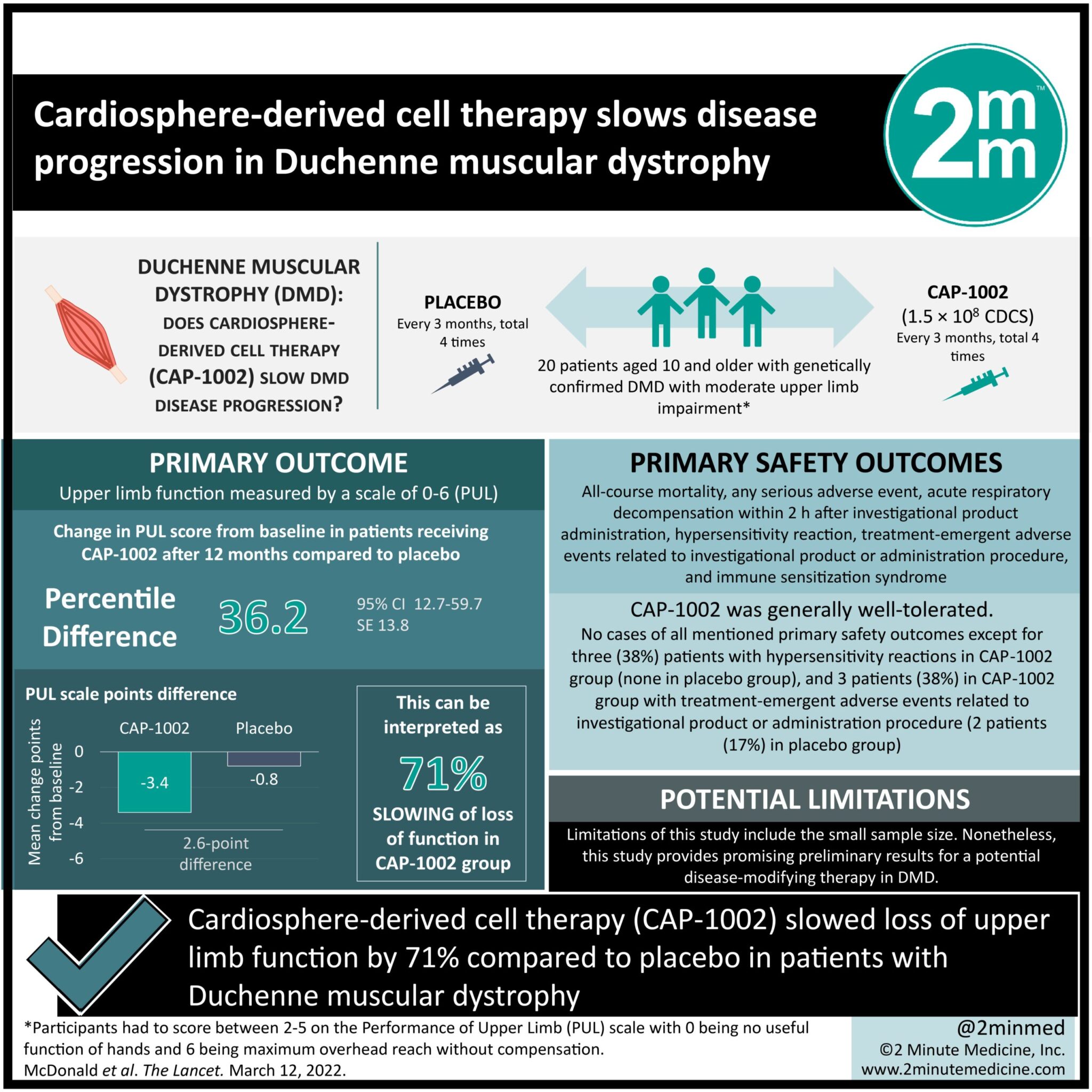
#VisualAbstract: Cardiosphere-derived cell therapy slows disease progression in Duchenne muscular dystrophy
1. Cardiosphere-derived cell therapy (CAP-1002) slowed loss of upper limb function by 71% compared to placebo in patients with Duchenne muscular dystroph
2. Treatment-emergent adverse events were rare, with one hypersensitivity allergic reaction occurring in the CAP-1002 group.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Duchenne muscular dystrophy (DMD) is an X-linked genetic disorder characterized by progressive muscle degeneration leading to significant reduction in life expectancy. Males with DMD have an estimated life expectancy of 22 years with heart and respiratory muscles affected in later disease stages. In this phase 2 trial, a formulation of allogenic cardiosphere-derived cells (CAP-1002) was evaluated against placebo in patients with DMD. CAP-1002 is, in essence, a concentrate of cardiac stem cells with potential disease-modifying properties such as regenerative abilities. Participants (n=20) were randomized 1:1 to receive either CAP-1002 or placebo every three months for four total infusions. Primary outcome was upper limb function measured by a scale of 0-6 (PUL). CAP-1002 was shown to slow PUL decline by 71% compared to placebo or by an absolute difference of 2.6 points. CAP-1002 was generally well-tolerated with only one severe adverse hypersensitivity reaction leading to withdrawal from the trial. Limitations of this study include the small sample size. Nonetheless, this study provides promising preliminary results for a potential disease-modifying therapy in DMD.
Click to read the study in the Lancet
Relevant Reading: Long-term effects of glucocorticoids on function, quality of life, and survival in patients with Duchenne muscular dystrophy: a prospective cohort study.
In-Depth [randomized controlled trial]: HOPE-2 was a randomized-controlled phase 2 clinical trial to assess to safety and efficacy of intravenous CAP-1002 for the treatment of Duchenne muscular dystrophy (DMD). The study enrolled patients aged 10 and older with genetically confirmed DMD. Participants had to score between 2-5 on the Performance of Upper Limb (PUL) scale with 0 being no useful function of hands and 6 being maximum overhead reach without compensation. 20 participants were assigned 1:1 to either CAP-1002 (n=8) or placebo (n=12) infusion every 3 months for a total of four infusions. Mean age of the enrolled male participants was 14 in both groups. Primary outcome was the upper limb function on the PUL scale. Patients who received CAP-1002 had a greater change in PUL score from baseline after 12 months compared to placebo (percentile difference 36.2, 95% CI 12.7-59.7). On the PUL scale, the placebo group had a mean change of -3.4 points from baseline, while the CAP-1002 had a -0.8 point change (difference of 2.6 points). This can also be interpreted as a 71% slowing of loss of function in the CAP-1002 group. Three patients in the CAP-1002 group had infusion-related hypersensitivity reactions, one leading to discontinuation. No other adverse events were seen in the two groups.
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.


![Gene therapy targets Duchenne muscular dystrophy in new trial [PreClinical]](https://www.2minutemedicine.com/wp-content/uploads/2018/10/70_lores-350x250.jpg)






