#VisualAbstract: Randomized Trial of Platelet-Transfusion Thresholds in Neonates
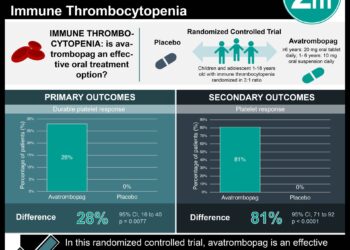
1. Preterm infants given prophylactic platelet transfusions at thresholds below 50,000 platelets per cubic millimeter had higher risks of death or major bleeding after 28 days as compared to neonates given transfusions at thresholds below 25,000 per cubic millimeter.
2. There was no significant difference in other serious adverse events between groups.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Preterm infants with thrombocytopenia are often given prophylactic platelet transfusions to reduce their risk of bleeding. However, guidelines for when to prophylactically begin platelet transfusions in preterm neonates with thrombocytopenia are currently lacking. In this randomized control trial, investigators studied the comparative effects of initiating platelet transfusions at platelet thresholds of 50,000 per cubic millimeter (high-threshold) versus 25,000 per cubic millimeter (low-threshold). The primary outcome was death or new major bleeding event within 28 days after randomization. Neonates randomized to receive platelet transfusions at below the higher-threshold had significantly higher death and major bleeding rates.
These findings may aid in creating objective threshold guidelines for platelet transfusions among neonates with thrombocytopenia. Limitations of the study include a minority of patients recruited during the first 5 days of life and possibly reduced generalizability as very few eligible patients were ultimately enrolled in the study.
Click to read the study in NEJM
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc