Zoledronic acid may increase bone density in frail elderly women
1. A single dose of zoledronic acid improved bone mineral density over 2 years in frail elderly women.
2. Rates of fracture or mortality did not differ significantly between zoledronic acid treatment and placebo groups.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Although as many as 85% of frail elderly adults living in long-term homes have osteoporosis, the current use of bisphosphonate therapy is low. Previous randomized controlled trials studying osteoporosis have not examined the efficacy of bisphosphonates in this population, and given physiological differences in the elderly, the efficacy and safety of bisphosphonates may be vastly different. This study aimed to determine the efficacy and safety of zoledronic acid to treat osteoporosis in frail elderly women living in long-term care facilities.
In this randomized controlled trial, a single dose of zoledronic acid improved bone mineral density in the population over 2 years. However, there was no significant decrease in rates of fracture or mortality across groups. Strengths of this study include studying this outcome in a novel yet important patient population. Limitations include the specific patient population selected, which may limit the generalizability of the results. Moreover, this study was not sufficiently powered to determine zoledronic acid’s effect on fracture rates, so further study regarding this question is warranted.
Click to read the study in JAMA Internal Medicine
Relevant Reading: Once-yearly Zoledronic acid for treatment of postmenopausal osteoporosis
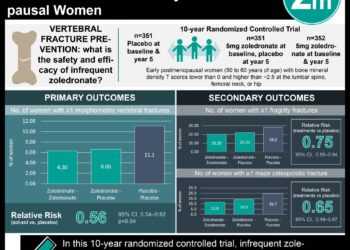
In-Depth [randomized controlled trial]: This double-blind, randomized controlled trial was performed in Pittsburgh, Pennsylvania from December 2007 to March 2012, and randomized a total of 181 frail elderly women to either a single dose of zoledronic acid 5 mg with vitamin D and calcium, or placebo (i.e., vitamin D and calcium alone). The primary outcome was hip and spine bone mineral density at 12 and 24 months. Secondary outcomes included rate of fracture and mortality. Data was analyzed using linear mixed model regression, χ2 and Fisher exact tests.
After randomization, the treatment group included more women with frailty, falls history, diabetes and anticonvulsant use. Women in the treatment group had a greater mean (standard error, SE) bone mineral density changes in the hip and spine at 24 months as compared to the placebo group: 2.6% (0.6%) vs. -1.5% (0.7%); p < 0.001 and 4.5% (0.8%) vs. 0.7% (0.5%); p < 0.001. Similar differences were also seen at the 12-month mark. Differences in fracture rates were not significant between groups – 20% for the treatment group and 16% for the placebo group (OR 1.30; 95%CI 0.61-2.78). Mortality rates also did not differ significantly across groups (OR 1.24; 95% CI 0.54-2.86).
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.



![Vaccines for predicted influenza strains may provide wide protection [PreClinical]](https://www.2minutemedicine.com/wp-content/uploads/2014/11/18156_lores-75x75.jpg)


