2 Minute Medicine Rewind March 28 – April 5, 2015
In this section, we highlight the key high-impact studies, updates, and analyses published in medicine during the past week.
Cell-free DNA Analysis for Noninvasive Examination of Trisomy
Cell-free DNA (cfDNA) is obtained from maternal blood samples and has been reported to have a high sensitivity for trisomy 21 and a low false negative rate and is thought to be a substantial improvement over previous screening methods involving nuchal translucency and biochemical analytes. However, to date there has yet to be a large-scale study of cfDNA compared to conventional screening methods in the general pregnancy population. In this prospective study, called the Noninvasive Examination of Trisomy (NEXT) study, investigators assigned 15,841 women, who presented for aneuploidy screening at 10 to 14 weeks of gestation, to undergo both standard screening and cfDNA testing. Researchers found that cfDNA testing detected 100% (95% CI 90.7-100) of trisomy 21 cases compared with 78.9% (62.7-90.4) of cases detected by standard screening. The cfDNA testing had a 0.06% (0.03-0.11) false positive rate compared to 5.4% (5.1 – 5.8) for the conventional screening method. Overall, the positive predictive value of cfDNA testing was 80.9% (66.7-90.0) compared with 3.4% (2.3-4.8) for standard screening (p < 0.001). This study therefore confirms that cfDNA is an effective method for trisomy 21 testing that has a higher sensitivity, lower false positive rate, and higher positive predictive value when compared with conventional screening methods that utilize nuchal translucency and biochemical analytes.
Pneumonia, sepsis, and meningitis are together responsible for approximately 700,000 neonate deaths each year. These diseases are often difficult to distinguish from one another by symptomology, which is why the WHO currently recommends that neonates with any clinical signs of possible serious bacterial infection are referred to tertiary care hospitals to receive intramuscular or intravenous antibiotic therapy. This strategy, however, is difficult to adhere to in low-income and middle-income countries, where tertiary care access is limited. In this study, investigators researched whether in comparison to intramuscular antibiotics, the use of an outpatient oral antibiotic regimen across five sites in Africa could be just as effective in treating neonates who displayed signs of rapid breathing, one of the most common signs of a serious bacterial infection. Researchers randomized 2333 infants aged 0-59 days with tachypnea as the only signs of a bacterial infection to receive either injectable procaine benzylpenicillin-gentamicin or oral amoxicillin. In the per-protocol analysis, where 91% of infants adhered to the injectable antibiotic regimen and 98% of infants adhered to the oral antibiotic regimen, this study found that 22% of patients in the injectable antibiotics group failed treatment (defined as clinical deterioration, development of a serious adverse event including death, persistence of fast breathing on day 4, or recurrence up to day 8) compared to 19% of infants in the oral amoxicillin group (risk difference -2.6%, 95% CI -6.0-0.8). This study therefore shows that young infants with fast breathing can be treated just as effectively with an outpatient oral amoxicillin regimen when referral to a hospital is not possible.
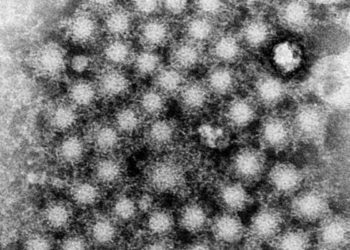
A Recombinant Vesicular Stomatitis Virus Ebola Vaccine — Preliminary Report
A number of vaccine candidates have been developed to address the recent Ebola epidemic concentrated in west Africa. The rVSV-ZEBOV vaccine is one especially promising vaccine candidate that utilizes an attenuated, replication-competent, recombinant vesicular stomatitis virus (rVSV) to express a Zaire strain of Ebola virus (ZEBOV) glycoprotein and has been shown to induce immunity in several non-human primates. In this phase 1 trial, 52 patients were randomized to receive either an injection of placebo, rVSV-ZEBOV at 3 million plaque-forming units (PFU), or rVSV-ZEBOV at 20 million PFUs. Investigators found that adverse events were minor and included injection-site pain, myalgia, and fatigue. All patients experienced transient VSV viremia. 28 days after vaccination, all individuals who had received the vaccine produced anti-ZEBOV glycoprotein antibodies as assessed by an enzyme-linked immunosorbent assay (ELISA). The group receiving the 20 million PFU dosage displayed a higher antibody titer compared to those receiving the 3 million PFU dose. This study therefore shows that the highly promising rVSV-ZEBOV vaccine has no identifiable major safety concerns and appears effective in inducing an anti-Ebola antibody response with greater effects seen at the higher vaccine dose.
New Hepatitis C virus (HCV) treatments have significant promise in curing viral infections at a higher rate while avoiding many of the toxic side effects of conventional interferon-based treatments. In this phase 3 randomized control trial, TURQUOISE-I, investigators studied the efficacy of an all-oral, interferon free, 3 direct-acting antiviral (3D) regimen consisting of ombitasvir, paritaprevir co-dosed with ritonanvir, dasabuvir, and ribavirin in patients co-infected with HCV genotype-1 and HIV-1. A total of 63 patients who were HCV treatment-naïve or had prior treatment failure and had HIV infection controlled on antiretrovirals were randomized to 12 or 24 weeks of the oral regimen. At 12 weeks post-treatment, sustained virologic response (HCV RNA < 25 IU/mL) was achieved in 94% (95% CI 79-98%) of patients who received the 12-week regimen and 91% (76-97%) of those receiving 24 weeks of treatment. The most common adverse effects were fatigue (48%), insomnia (19%), nausea (18%), and headache (16%). This study therefore showed that in patients co-infected with HCV-1 and HIV-1, a regimen consisting of ombitasvir, paritaprevir/ritonavir, dasabuvir, and ribavirin was highly effective in producing a sustained virologic response with similar effects after 12 or 24 weeks of treatment and with minor adverse effects.
Impact of Statins on Serial Coronary Calcification During Atheroma Progression and Regression
The value of cardiac computed tomography (CT) in visualizing and identifying coronary artery calcification is uncertain as it is not known whether plaque volume, location, and morphology are the optimal metrics to predict clinical outcomes. Statins are a mainstay of the medical treatment of coronary artery disease and have been suggested to have in-vitro plaque stabilizing effects. In this retrospective analysis of 3495 patients from 8 studies, investigators reviewed the use of statins (high vs. low intensity statins vs. none) and correlated cardiac CT findings including coronary artery calcification. High intensity statin therapy was associated with more atheroma volume regression from baseline compared to low intensity statin therapy (-0.6% vs. 0.8%, p < 0.001) and no statin therapy (-0.6% vs. 1.0%, p < 0.001). There were increased in calcium from baseline in all groups, which could potentially stabilize plaques. Statins appear to both cause plaque regression as well as increase calcification of plaques, which could lead to fewer clinical events of plaque rupture.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.





![Small molecule drug may improve cancer-killing efficacy of oncolytic virus [PreClinical]](https://www.2minutemedicine.com/wp-content/uploads/2015/04/Microtubules_in_the_leading_edge_of_a_cell.tif-75x75.jpg)


