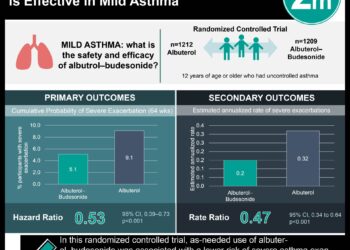
Albuterol-budesonide combination efficacious for asthma exacerbation rescue
1. A high-dose combination inhaler of albuterol and budesonide was superior to albuterol alone in reducing the risk of severe asthma exacerbation.
2. Albuterol-budesonide combination had a similar safety profile to albuterol-alone treatment.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Asthma is a chronic disease that involves airway hypersensitivity, inflammation, and obstruction. Glucocorticoids, such as budesonide, serve as maintenance therapy. Though, patients experiencing loss of asthma control also rely on albuterol and other short-acting β2-agonists (SABAs) for rescue. It is of concern that in uncontrolled asthma rescue SABA alone does not address underlying inflammation and carries a risk of severe exacerbation. Fixed-dose combinations of glucocorticoid and SABA are being explored as alternative rescue therapies for this population. The present study compared the efficacy of fixed-dose combination regimens of budesonide at high and low doses with albuterol against albuterol alone in preventing severe asthma exacerbation in high-risk patients. Patients continued to receive glucocorticoid-containing maintenance therapy throughout the trial. It was found that the combination of albuterol and high-dose budesonide resulted in a significantly lower risk of severe exacerbation compared to albuterol alone. The incidence of adverse events was similar across groups. Although children only comprised a minority in this trial where statistical significance was lacking, the results provided evidence to support the use of budesonide-albuterol combination as rescue therapy for asthma patients at risk of severe exacerbation.
Click here to read the study in NEJM
Relevant Reading: Rescue use of beclomethasone and albuterol in a single inhaler for mild asthma
In-Depth [randomized control trial]: The present double-blind randomized control trial compared the efficacy of albuterol-budesonide combinations against albuterol alone as rescue therapy for patients with poorly controlled high-risk asthma. Patients four years and older with symptomatic asthma who had had at least one severe exacerbation in the previous 12 months were recruited. Patients were randomized in a 1:1:1 ratio to one of three inhaler regimens: a higher-dose combination of 180µg albuterol and 160µg budesonide, a lower-dose combination of 180µg albuterol and 80µg budesonide, or 180µg albuterol alone. The primary efficacy endpoint was the time to first event of severe asthma exacerbation. Secondary efficacy endpoints included annualized rate of exacerbations, total systemic glucocorticoid need, and quality of life questionnaires at week 24. The risk of severe asthma exacerbation was 26% lower in the high-dose combination group than albuterol-alone (hazard ratio [HR], 0.74; 95% confidence interval [CI], 0.62 to 0.89; p=0.001). The HR in the lower-dose combination against albuterol-alone was 0.84 (95% CI, 0.71 to 1.00; p=0.052). The annualized rate of severe exacerbations was 0.43 (95% CI, 0.33 to 0.58) for the high-dose combination and 0.58 (95% CI, 0.44 to 0.77) for albuterol-alone (rate ratio (RR), 0.75; 95% CI, 0.61 to 0.91). The annualized rate of severe exacerbations was 0.48 (95% CI, 0.37 to 0.63) for lower-dose combination and 0.60 (95% CI, 0.46 to .079) for albuterol alone (RR, 0.81; 95% CI, 0.66 to 0.98). The higher-dose combination was also associated with improvements in quality of life. Adverse event rates were similar across the three groups. Overall, the study results added to the current body of evidence supporting the combination of glucocorticoids and β2-agonists as rescue therapy to reduce the risk of severe exacerbations in patients with poorly controlled asthma.
Image: PD
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.




![The ABCD2 score: Risk of stroke after Transient Ischemic Attack (TIA) [Classics Series]](https://www.2minutemedicine.com/wp-content/uploads/2013/05/web-cover-classics-with-logo-medicine-BW-small-jpg-75x75.jpg)