Atezolizumab may improve disease-free survival in mismatch repair deficiency locally advanced colon cancer
1. In patients with resected stage III mismatch repair-deficient colon cancer, adjuvant atezolizumab plus standard chemotherapy improved disease-free survival compared to chemotherapy alone.
2. The combination regimen was associated with higher rates of adverse events, though these were consistent with known toxicity profiles.
Evidence Rating Level: 1 (Excellent)
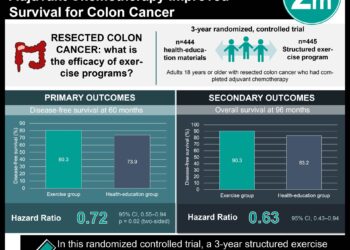
Study Rundown: Colorectal cancer remains a leading cause of cancer-related mortality worldwide, and a subset of tumors characterized by mismatch repair deficiency (dMMR) demonstrates distinct biology, including high immunogenicity and responsiveness to immune checkpoint blockade. While immunotherapy has become standard in metastatic dMMR disease, its role in earlier-stage, curative settings has been less clear. This randomized controlled trial evaluated whether adding atezolizumab, a programmed death ligand 1 (PD-L1) inhibitor, to standard adjuvant chemotherapy (fluorouracil, oxaliplatin, and leucovorin known as mFOLFOX6) would improve outcomes in patients with resected stage III dMMR colon cancer. It was found that the addition of immunotherapy led to a clinically meaningful improvement in disease-free survival, suggesting enhanced eradication of micrometastatic disease. However, overall survival data were immature, and no clear benefit was observed in that endpoint. Strengths of the study include its randomized design, broad multicenter enrollment, and focus on a specific biologically defined subgroup likely to benefit from immunotherapy. Limitations include relatively short follow-up for overall survival, potential confounding from subsequent immunotherapy at recurrence, and increased toxicity with combination therapy. Additionally, the trial design did not allow for the assessment of the independent contribution of immunotherapy apart from chemotherapy. Nonetheless, these results suggest that the addition of atezolizumab to standard therapy may enhance disease-free survival among patients with resected stage III mismatch repair deficient colon cancer.
Click to read the study in NEJM
Relevant Reading: Defective mismatch repair as a predictive marker for lack of efficacy of fluoro- uracil-based adjuvant therapy in colon cancer.
In-Depth [randomized controlled trial]: This phase 3 randomized controlled trial (ATOMIC) evaluated the addition of atezolizumab, a PD-L1 inhibitor, to standard adjuvant mFOLFOX6 chemotherapy in patients with resected stage III mismatch repair–deficient (dMMR) colon cancer. A total of 712 patients were randomized in a 1:1 ratio to receive either atezolizumab plus mFOLFOX6 for 6 months followed by atezolizumab monotherapy (total 12 months), or mFOLFOX6 alone for 6 months. Baseline characteristics were well balanced between groups, including a substantial proportion of high-risk tumors (T4 and/or N2 disease). At a median follow-up of 40.9 months, those who received atezolizumab had significantly improved disease-free survival compared to those who received chemotherapy alone (86.3% versus 76.2%; hazard ratio for recurrence or death, 0.50; 95% confidence interval [CI], 0.35 to 0.73; p<0.001). This finding was consistent across most prespecified subgroups, including nodal status, tumor stage, and performance status. An exploratory analysis suggested that patients receiving more than 6 cycles of chemotherapy derived greater benefit (HR 0.41), whereas those receiving fewer cycles did not (HR 0.97). At the time of analysis, overall survival was similar between groups (HR 0.90, 95% CI 0.55–1.47; p=0.68). Notably, many patients in both arms received immunotherapy upon recurrence, which may have attenuated OS differences. Safety analysis showed higher rates of grade 3–4 adverse events in the atezolizumab group (84.1% vs. 71.9%), most commonly neutropenia (43.6% vs. 35.9%) and fatigue (10.1% vs. 3.3%). Overall, this study demonstrated that atezolizumab may offer a benefit in disease-free survival among patients undergoing adjuvant chemotherapy for stage III mismatch repair deficient colon cancer.
Image: PD
©2026 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.