Azithromycin less effective compared to doxycycline for chlamydial infections
1. Non-inferiority of azithromycin was not established as compared to doxycycline in treating chlamydial infections, likely due to the 100% efficacy of doxycycline, as the efficacy was 97% for azithromycin.
2. However, given the closed population with direct observed treatment and per-protocol primary analysis, it is not clear if the efficacy of doxycycline would remain as high in practice.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Chlamydia trachomatis is a sexually transmitted infection that can cause serious sequelae like pelvic inflammatory disease. Current treatment recommendations include a onetime dose of 1gram azithromycin or doxycycline 100 mg twice daily for 7 days. At the time of these recommendations, it was thought that azithromycin and doxycycline were equi-efficacious however with studies using new nucleic acid amplification tests (NAAT), which are more sensitive to treatment failure, the efficacy of azithromycin has been called into question. This study compared oral azithromycin with doxycycline in treating urogenital chlamydia infection to evaluate non-inferiority of standard azithromycin to standard doxycycline treatment.
Non-inferiority of azithromycin to doxycycline could not be established in this study. There were no treatment failures in those randomized to doxycycline however treatment failure occurred in 3.2% of the azithromycin treatment group. Overall, the efficacy of doxycycline was 100% while azithromycin was 97%. A strength of this study includes using a setting where medication adherence would not be a contributing factor. However, when treatment is provided in the real world, there is a possibility that twice-daily doxycycline dosing may result in decreased adherence and efficacy comparisons in this circumstance were not assessed in this study.
Click to read the study, published today in NEJM
Relevant Reading: Azithromycin versus doxycycline for genital chlamydial infections: a meta-analysis of randomized clinical trials
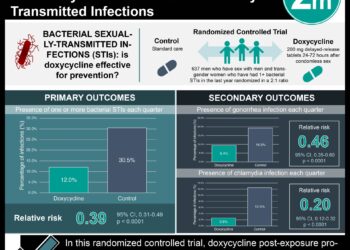
In-Depth [randomized controlled trial]: This per-protocol, randomized-controlled trial was conducted in four long-term youth correctional facilities in Los Angeles, USA from December 2009 to May 2014. Inclusion criteria were any correctional resident with a positive screening NAAT on intake. Exclusion criteria included residents who were pregnancy, breast-feeding, had a gonorrhea co-infection among others. Residents were randomized 1:1 to either standard treatment of either azithromycin or doxycycline. The primary outcome of interest was treatment failure, determined by NAAT and C. trachomatis genotyping, at 28 days after treatment. Non-inferiority would be determined if the absolute rate of azithromycin treatment failure would be less than 5% higher than the absolute rate of doxycycline treatment failure.
The 567 participants were randomized 284 to azithromycin and 283 to doxycycline. However, due to numerous reasons, primarily because of discharge from the facility, only 155 residents were included in each primary evaluation. No treatment failures occurred in the doxycycline group (0%; 95% [CI] 0.0% to 2.4%). Five participants (four male and one female) in the azithromycin group had treatment failure (3.2%; 95% [CI] 0.4% to 7.4%). The difference in failure rate between the two treatments was 3.2% (one-sided 90% [CI], 0.0 to 5.9%). The upper limit of the 90% [CI] was outside of the predetermined 5% rate difference and thus non-inferiority of azithromycin to doxycycline could not be established.
Image: PD/CDC
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.