Combination chemotherapy associated with improved survival in refractory colorectal cancer [RECOURSE]
1. TAS-102 is a combination of a thymidine-based nucleic acid analogue, trifluridine, and a thymidine phosphorylase inhibitor, tipiracil hydrochloride.
2. Patients with refractory colorectal cancer treated with TAS-102 had significantly improved median overall survival compared to patients treated with placebo; neutropenia and leukopenia were seen in 38% and 21% of the TAS-102 treated patients, respectively.
Evidence Rating Level: 1 (Excellent)
Study Rundown: The contemporary standard chemotherapy regimen for metastatic colorectal cancer (CRC) is a combination of fluorouracil and leukovorin, irinotecan or oxaliplatin, and either bevacizumab or an epidermal growth factor inhibitor. Ongoing research seeks to elucidate agents that can serve in cases of metastatic CRC that are refractory despite these treatments. One such agent is TAS-102, a combination of a thymidine-based nucleic acid analogue, trifluridine, and a thymidine phosphorylase inhibitor, tipiracil hydrochloride. In previous phase 1 and 2 clinical studies, this agent showed promise in the treatment of refractory CRC.
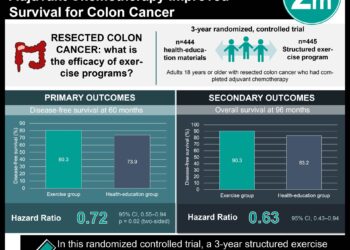
In this multinational, phase 3 randomized placebo-controlled trial, 800 patients with metastatic CRC refractory to standard therapies received TAS-102 or placebo. Patients had previously undergone treatment within multiple rounds of standard chemotherapies or had experienced clinically significant side effects from standard therapy that precluded being treated again. In a survival analysis, the median overall survival in the TAS-102 group was 7.1 months compared to 5.3 months in the placebo group; a significant improvement was also noted in overall progression-free survival and median time to worsening performance status. Common adverse effects associated with treatment with TAS-102 included neutropenia (38%) and leukopenia (21%); febrile neutropenia occurred in 4% of patients treated with TAS-102 and was associated with death due to sepsis in one case.
This drug-company-sponsored randomized, controlled trial provides solid grounding that TAS-102 provides benefit in the treatment of refractory CRC, though treatment must be weighed against the associated side effect profile. Of note, this finding was consistent across patients regardless of KRAS mutation tumor status, but insufficient tumor specimens were assessed for BRAF status to test within this subgroup.
Click to read the study, published today in NEJM
Relevant Reading: Systemic Therapy for Colorectal Cancer
In-Depth [randomized controlled trial]: This multinational phase 3 randomized, placebo-controlled trial involved 800 patients with metastatic CRC refractory to standard therapies who received TAS-102 or placebo. Patients were required to have adequate bone-marrow, liver, and renal function, and to have a Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 (indicating either no symptoms or mild symptoms). Patients were randomized in a 2:1 ratio to receive TAS-102 or placebo. In the primary outcome analysis, patients with TAS-102 had a median overall survival of 7.1 months compared to 5.3 months in the placebo group (HR for death, 0.68, 95% CI, 0.58 to 0.81, P<0.001); this finding was consistent across almost all subgroup analyses. The hazard ratio for progression or death for TAS-102 versus placebo was 0.48 (95%CI, 0.41-0.57, P<0.001), which was also consistent across subgroup analyses (including KRAS genotype, age, geographic region, gender, ECOG performance status, and prior treatment characteristics). Serious adverse events occurred in 30% of those treated with TAS-102 compared to 34% of those in the placebo arm. 38% of those receiving TAS-102 experienced neutropenia of grade 3 or higher, 4% had febrile neutropenia (1 case associated with sepsis and death), and 9% received granulocyte colony-stimulating factor.
Image: PD
©2015 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.


![Weakened grip strength associated with increased morbidity and mortality [PURE study]](https://www.2minutemedicine.com/wp-content/uploads/2015/05/9266_lores1-75x75.jpg)


