Dolutegravir regimen more effective than standard regimen in treatment of HIV-1
Image: PD
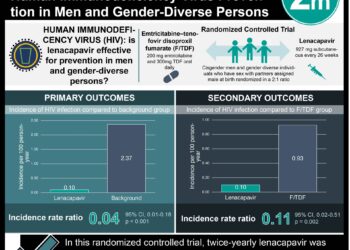
1. At 48 weeks, the proportion of patients with an HIV-1 RNA level of less than 50 copies/ml was significantly higher in the dolutegravir regimen group than the standard single-tablet regimen group.
2. Fewer patients in the dolutegravir group discontinued therapy due to adverse events compared to the standard regimen group.
Evidence Rating Level: 1 (Excellent)
Study Rundown: In this randomized, double-blind trial involving individuals with HIV-1 who had not previously received therapy, the authors showed that a single-tablet regimen consisting of the integrase inhibitor dolutegravir, plus abacavir-lamivudine (DTG-ABC-3TC) was more effective than fixed-dose efavirenz-tenofovir disoproxil fumarate (DF)-emtricitabine (EFV-TDF-FTC), the only single-tablet regimen currently preferred in U.S. HIV treatment guidelines. Effectiveness was scored primarily in terms of the proportion of patients reaching a threshold of virologic suppression (HIV-1 RNA level of less than 50 copies per milliliter) at 48 weeks. Dolutegravir is an unboosted integrase inhibitor with a long plasma half-life supporting once-daily dosing. Its potential advantages include the fact that it does not affect the cytochrome P-450 enzyme CYP3A4 (fewer drug interactions) and has activity against viruses that are resistant to nonnucleoside reverse-transriptase inhibitors.
The study’s findings suggest that dolutegravir could potentially be added to ongoing multiple antiretroviral class regimens in a single-tablet with an effectiveness that is on a par with—and potentially superior to—existing single-tablet regimens. Strengths of the study include its randomized, double-blinded design and the high level of compliance with the study treatment. Limitations include the fact that only 16% of the participants were women, and there was a relatively low proportion of participants with a CD4+ T-cell count of less than 200 per cubic millimeter. Furthermore, long-term studies will have to follow these subjects to ensure that the effectiveness and acceptable adverse effect profile persist beyond the relatively short study period of 48 weeks. Finally, the study was supported by ViiV healthcare, which is the producer of dolutegravir.
Click to read the study, published today in NEJM
Relevant Reading: Once-daily dolutegravir versus raltegravir in antiretroviral-naïve adults with HIV-1 infection
In-Depth [randomized controlled trial]: This trial randomly assigned 833 participants to receive one of two drug combinations: DTG-ABC-3TC or EFV-TDF-FTC. Eligible participants were 18 years or older, had a plasma HIV-1 RNA level of at least 1000 copies per milliliter without genotypic evidence of viral resistance at screening, and were treatment-naive. The primary efficacy end point was the proportion of participants with a plasma HIV-1 RNA level of less than 50 copies per milliliter at week 48. Secondary efficacy end points included the time to viral suppression and the change from baseline in CD4+ T-cell count. Participants were also monitored for adverse events related to drug administration and the emergence of viral resistance to drug regimens.
In the intention-to-treat analysis, 88% of participants in the DTG-ABC-3TC group achieved the primary end point of plasma HIV-1 RNA level of less than 50 copies per milliliter at week 48, versus 81% of those in the EFC-TDF-FTC group (adjusted treatment difference, 7 percentage points; 95% CI, 2 to 12). This difference of 7% was observed among participants with a high baseline HIV-1 RNA level (>100,000 copies per milliliter) as well as those with a low baseline HIV-1 RNA level; it was present across demographic subgroups including race, sex, and age. Overall differences in response were due primarily to adverse events leading to discontinuations (2% participants in the DTG-ABC-3TC group, with diarrhea and insomnia being most common causes, versus 10% in the EFV-TDF-FTC group). The median time to viral suppression was 28 days in the DTG-ABC-3TC group versus 84 days in the EFV-TDF-FTC group (P<0.001).
Adverse events of grade 3 or 4 were reported in 10% of the DTG-ABC-3TC group versus 16% of the EFV-TDF-FTC group. Patients in the DTG-ABC-3TC group had small mean increases in serum creatinine levels, evident by week 2 and stable through week 48; no change from baseline was detected in the urinary albumin-to-creatinine ratio. Viral resistance did not develop during treatment in any patients in the DTG-ABC-3TC group.
By Matthew Growdon and Adrienne Cheung
More from this author: The CHEST-1 & PATENT-1: Riociguat significantly improves exercise capacity in pulmonary hypertension patients, High glucose levels associated with increased risk of dementia, Insecticidal bed nets linked with reduced transmission of lymphatic filariasis, Fecal occult-blood testing linked with reduction in colorectal-cancer mortality, High-flow nasal cannulae noninferior to nasal CPAP in very preterm infants after extubation
©2012-2013 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.