Erythropoietin linked with reduced risk of brain injury in preterm infants
1. The administration of high dose recombinant erythropoietin to premature infants within 42 hours after birth was associated with a decreased risk of brain injury.
2. Analysis of the primary outcome of this study assessing brain development at 2 years of age is pending.
Evidence Rating Level: 2 (Good)
Study Rundown: Protecting and promoting the neurological development of preterm infants is a challenging task. Erythropoietin, a hormone known to control red blood cell production, has been additionally shown to demonstrate a protective effect on the nervous system of animals. The purpose of this study was to investigate the effects of high dose recombinant human erythropoietin on the neural development of preterm infants. Data gathered using T1 and T2 weighted MRI images of preterm infants was compared to MRI studies from age-matched controls and analyzed for evidence of brain injury. While a lack of high quality MRI access to all patients posed a study limitation, erythropoietin given within 42 hours of birth was associated with a decreased risk of brain injury to both the white and gray matter of the brain. Analysis of the primary outcome of this study, brain development at 2 years of age, is pending.
Click to read the study, published today in JAMA
Relevant Reading: Cognitive Outcomes of Preterm Infants Randomized to Darbepoetin, Erythropoietin, or Placebo
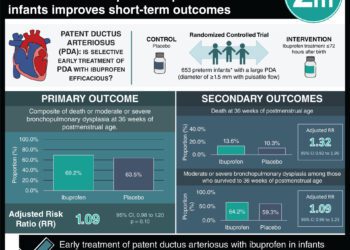
In-Depth [randomized controlled trial]: This study evaluated 165 infants from Switzerland between 2005 and 2012 (n=77 erythropoietin; n=88 placebo). A statistically significant association was observed between erythropoietin administration and improved white matter injury (WMI) scores (mean score, 5.91 [SD, 1.33] for recombinant human erythropoietin and 6.43 [SD, 1.76] for placebo; mean difference, −0.52; 95% CI, -1.01 to -0.04; P = .04). Erythropoietin treatment was also associated with significantly fewer abnormal gray matter injury (GMI) scores in the recombinant human erythropoietin group (7%) compared with the placebo group (19%) (difference, 12%; 95% CI, 2%-22%; P =.03).
More from this author: Reducing surgical complications may increase costs, Protected sleep periods improve intern alertness and sleep duration, ADHD medication decreases rates of criminality in ADHD patients, Low dose aspirin shows net clinical benefit in patients with first unprovoked venous thromboembolism, Rare TREM-2 mutation implicated in Alzheimer’s Disease
Image: PD
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.




![Macitentan better than placebo for preventing progression of pulmonary arterial hypertension [SERAPHIN Trial]](https://www.2minutemedicine.com/wp-content/uploads/2013/08/ca68dc0329bef0b42ccae7b6656a0f_gallery-75x75.jpg)