Glyburide is not non-inferior to insulin in reducing perinatal complications in gestational diabetes
1. In this randomized controlled trial, glyburide was not non-inferior in terms of the composite rate of macrosomia, neonatal hypoglycemia, and hyperbilirubinemia compared with subcutaneous insulin.
2. Women taking glyburide had better glycemic control and were more likely to prefer glyburide for subsequent pregnancies but also had higher rates of maternal and neonatal hypoglycemia.
Evidence Rating Level: 1 (Excellent)
Study Rundown: There is an increasing risk of gestational diabetes worldwide, with adequate treatment reducing fetal and maternal morbidity. While insulin remains first-line, glyburide has also been used due to its convenience as an oral drug, though it is unclear if glyburide has similar efficacy to insulin. The current study failed to demonstrate non-inferiority for glyburide compared to subcutaneous insulin for the prevention of overall perinatal complications, including macrosomia, neonatal hypoglycemia, and hyperbilirubinemia. In terms of individual endpoints, a greater rate of hypoglycemia was detected for glyburide. Women taking glyburide had better fasting glycemic control and were more likely to prefer the same treatment for subsequent pregnancies but also displayed higher rates of severe hypoglycemia.
Though this is the first study to use neonatal criteria as a primary outcome, it remains unclear if glyburide is non-inferior to subcutaneous insulin. While the higher rates of neonatal hypoglycemia with glyburide were clear, a non-significant increase in macrosomia for those treated with insulin should also be noted. In addition, there were huge divides in satisfaction between the treatment regimens, and this study was unable to determine the risk-benefit of an oral regimen in glyburide over subcutaneous insulin injections. A randomized, pragmatic study design may be beneficial in the future to determine what the “real-world” rates of complications would be between these two treatment strategies.
Click here to read the study, published in JAMA
Relevant Reading: Trends in Glyburide Compared With Insulin Use for Gestational Diabetes Treatment in the United States, 2000–2011
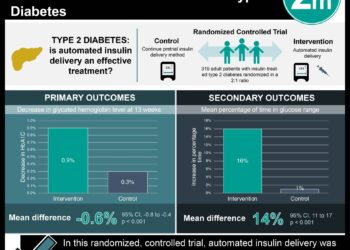
In-Depth [randomized control trial]: This multicenter, randomized, non-inferiority trial recruited 914 women (367 assigned to glyburide and 442 assigned to subcutaneous insulin) with a singleton pregnancy who were diagnosed with gestational diabetes between 24 and 34 weeks of gestation. The primary outcome was a composite of perinatal complications that included macrosomia, neonatal hypoglycemia, and hyperbilirubinemia. Overall, the frequency of the composite score for glyburide did not meet non-inferiority criteria, with a 27.6% complication rate in the glyburide group and 23.4% in the subcutaneous insulin group (p = 0.19). Post-hoc analysis revealed a higher rate of neonatal hypoglycemia with glyburide (difference 5%; CI95 0.5-9.5%) and no significant difference in macrosomia (-1.4%; CI95 -6.6 to 3.4%) or hyperbilirubinemia (-0.6%; CI95 -3.3 to 2.0). More women with glyburide had good fasting glycemic control than insulin (8.5%; CI95 1.9 to 15.2%) but also had higher rates of hypoglycemia (difference, 25.3%; CI95 16.6 to 34.0%) and were more likely to have a severe hypoglycemic episode (2.8%; CI95 0.2 to 5.5%).
Image: PD
©2018 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.