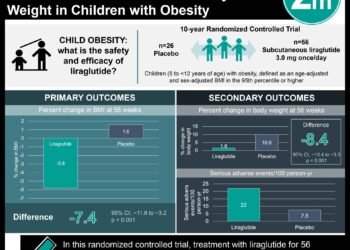
Metformin linked to decreased BMI z scores in prepubertal children
1. Prepubertal obese children treated with metformin over 6 months had significantly lower BMI z scores compared to those receiving placebo. These differences were not present in pubertal children.
2. Prepubertal obese children treated with metformin over 6 months had higher qualitative insulin-sensitivity check index (QUICKI), or lower rates of insulin resistance, compared to those receiving placebo. Additionally, prepubertal obese children had lower concentrations of INF-gamma and tPAI-1 at 6 months compared to prepubertal children on placebo.
Evidence Rating: 1 (Excellent)
Study Rundown: Obesity and diabetes are an increasing problem in the pediatric population. Metformin is an established oral antihyperglycemic agent approved for the treatment of adults with type 2 diabetes mellitus and in children >10 years of age. In adults, metformin has been shown to significantly reduce obesity. Similar existing trials in pediatric patients have not taken into consideration the stages of puberty as a confounding factor in the effects of metformin on weight loss. In this study, researchers investigated whether treatment with oral metformin is capable of reducing BMI z score, insulin sensitivity and inflammatory biomarkers in obese children, while stratifying for pubertal stage by Tanner staging. Researchers found an exclusive significant decrease in BMI z score in prepubertal children in the metformin group compared to the placebo group at 6 months. Additionally, prepubertal children in the metformin group had significantly lower scores of insulin resistance and lower concentrations of pro-inflammatory markers, including INF-gamma and tPAI-1. The authors suggest that the lack of significant results observed in the pubertal group may be due to the well-known decrease in insulin sensitivity known to occur in this population. Limitations of the study include medication compliance assessment by pill bottle return, as opposed to pill counting, and difficulty in assessing the effects of lifestyle changes in the children throughout the study. These results suggest that while metformin offers several metabolic benefits in prepubertal children, puberty itself may impact the extent of these effects.
Click to read the study, published today in Pediatrics
Relevant Reading: Metformin and body weight
In-Depth [randomized controlled trial]: Participants in this randomized, double-blind, placebo-control trial included 140 children (82 boys, 68 girls) across 4 Spanish hospitals. The primary outcome was BMI z score compared to baseline after 6 months. Secondary outcomes included insulin resistance and concentrations of inflammatory markers. In the prepubertal population, BMI z scores were significantly decreased in the metformin group when compared to placebo (P = 0.04). Baseline and 6 month BMI z scores were 4.0 ± 0.2 and 3.4 ± 0.2 for the placebo group, and 3.4 ± 0.2 and 2.6 ± 0.20 for the metformin group. Such significant differences were not observed in the pubertal group. Additionally, binary logistic regression showed an independent association between BMI z score and metformin treatment (OR = 0.18 [95% CI = 0.050-0.636; P = 0.01]. Insulin sensitivity was determined using the Qualitative insulin-sensitivity check index (QUICKI) – a measure of insulin resistance based on fasting glucose and insulin levels, with lower levels indicating resistance. Prepubertal children in the metformin group had significantly increased QUICKI scores compared to those receiving placebo (P = 0.01). No significant differences were seen in the pubertal group. Of the inflammatory markers tested, prepubertal children had lower concentrations of INF-gamma (P = 0.02) and tPAI-1 (P = 0.04). Again, these significant differences were not seen in pubertal children.
©2017 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.