Physician notification increases patient participation in colorectal cancer screening
1. Providing physicians with a list of patients requiring colorectal cancer screening results in a small but significant increase in patient participation in fecal immunochemical test cancer screening.
Evidence Rating Level: 1 (Excellent)
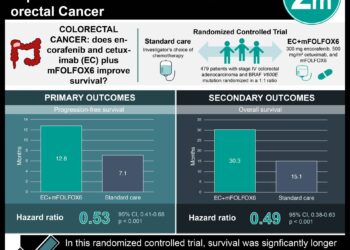
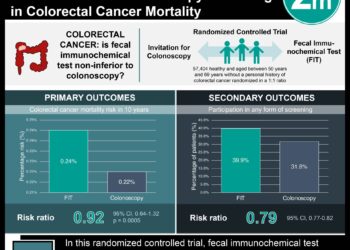
Study Rundown: Colorectal cancer is the third most diagnosed cancer in the world and the second most lethal after lung cancer. Endoscopic tests such as colonoscopy or a flexible sigmoidoscopy, and stool based tests such as the fecal immunochemical test (FIT) or the fecal occult blood tests are common screening tests that are used in early diagnosis of the disease. The present study is a randomized clinical trial that was conducted in France and investigated if providing a list of nonadherent patients to physicians improved patient participation in FIT colorectal cancer screening. In total, 1482 physicians participated and randomized into three groups based on the information they received: patient specific reminders group (list of patients that had not undergone colorectal cancer screening), generic reminders group (region-specific colorectal cancer screening rates), and usual care group (no reminders at all). A total of 33 044 patients were included in this study. At 1 year, there was a 3.1% difference between the patient-specific reminders group and the generic-reminders group, and 4.2% difference between the patient-specific reminders group and the usual care group.
Overall, the results of this study suggest that providing physicians with patient specific reminders on colorectal screening has the effect of increasing patient participation in FIT screening. The study is limited by the small magnitude of the observed increase in the population as well as difficulty in discerning what additional factors made the increased participation in the patient specific reminders population possible.
Click to read the study, published in JAMA
Relevant Reading: Screening for Colorectal Cancer and Evolving Issues for Physicians and Patients: A Review
In-Depth [randomized clinical trial]: This study is a cluster randomized control trial that consisted of 3 parallel groups, and was conducted on the French west coast from July 14, 2015, to July 14, 2016. General practitioners in 2 areas on the French west coast were eligible if they had at least 100 patients older than 16 years on their patient list. Since 2015, FIT test has been used more commonly in France. As part of the colorectal cancer screening program in France, local health officials invite patients to participate in the screening if they are eligible and have not completed their FIT screening in the preceding 24 months. These patients may typically receive an FIT screening kit at their local general practitioner’s office, which they can then return for screening purposes. Patients with a positive screening result would result in a referral to a gastroenterologist for endoscopic testing. Patients who do not receive their FIT screening within 3 months are labeled as non-adherent and these patients may consequently receive a reminder letter at 3 months and then again at 6 months. This study focused on the average-risk patients at a general practitioner practice that had not completed FIT testing within the 3 months of receiving their first invitation. The primary end point of the study was patient participation in the colorectal cancer screening program 1 year after the initial 3-month reminder had been mailed. Screening participation rates based on patient characteristics comprised the secondary endpoints. Overall, 1482 physicians (mean age, 53.4 years; 576 women [38.9%]) were randomized in this study into the three groups; patient-specific reminders, generic reminders, and usual-care groups, that consisted of 33 044 patients (mean age, 59.7 years; 17 949 women [54.3%]). Providing physicians with specific reminders about patients needing colorectal cancer screening improved patient participation in the FIT cancer screening.
Image: CC/Wiki
©2017 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.