Recombinant human pentraxin 2 slows the decline in lung function for patients with idiopathic pulmonary fibrosis
1. The use of recombinant human pentraxin 2 vs placebo resulted in a slower decline in lung function over 28 weeks for patients with idiopathic pulmonary fibrosis.
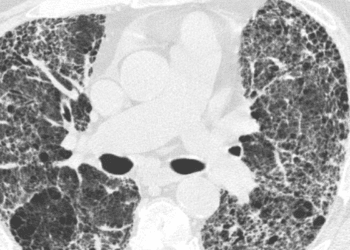
2. No significant treatment differences were observed in total lung volume, parenchymal features on high resolution CT, or diffusion capacity of carbon monoxide (DLCO).
Evidence Rating: 1 (Excellent)
Study Rundown: Idiopathic pulmonary fibrosis (IPF) is a progressive disease that leads to an irreversible loss of lung function and has a 5-year survival of 20-40%. Current therapies including pirfenidone and nintedanib do slow the rate of decline in forced vital capacity (FVC) but do not halt disease progression. However, this study, by building on previous research showed that human recombinant pentraxin 2, a known potent inhibitor of pro-inflammatory changes leading to IPF, reduces the decline in FVC percentage of predicted value as well as 6-minute walk distance.
A significant treatment effect for recombinant human pentraxin 2 vs placebo on the primary end pint of FVC change was observed. However, there were a few limitations to this study. First, the sample size used to demonstrate superiority of treatment over placebo was not appropriate to explore additional hypothesis beyond the pre-specified primary analysis and secondary end point should be considered exploratory. Second, the diagnosis of IPF was made in accordance with current guidelines, but allowing for possible usual interstitial pneumonia, which has been reported to be radiologic subtype present in substantial fraction of patients with IPF. Third, high resolution CT images were not read centrally, introducing heterogeneity into the patient population.
Click to read the study in JAMA
Relevant Reading: Pentraxin 2 in healthy volunteers versus patients with pulmonary fibrosis
In-Depth [randomized clinical trial]: During the study period from August 2015 to May 2017, 117 patients underwent randomization (81% male, mean age 68.6 years, mean time since IPF diagnosis 3.8 years). Eligible patients were between the ages of 40 and 80 years with a diagnosis of IPF, FVC of 50% or greater and of 90% or less predicted, DLCO of 25% or greater and 90% or less predicted, distance of at least 150 m on the 6-minute walk test, and ratio of forced expiratory volume in 1 second (FEV1)/FVC greater than 0.70. Exclusion criteria including significant emphysema, cigarette smoking within the past 3 months, or oxygen saturation by pulse oximetry of 89% or less on room air. 91 patients were receiving concurrent therapy (pirfenidone or nintedanib) at baseline. The primary end point showed a -2.5% change in treatment group compared with a -4.8% change in placebo for FVC percentage after therapy (p=0.001, [CI90 1.1-3.5]). No significant differences in least –squares mean changes were observed in the recombinant human pentraxin 2 group compared with the placebo group from baseline to week 28 for total lung volume (93.5 mL [CI90 -27.7-214.7; p = 0.20]), percentage of normal lung volume (-1.2% [CI90 -4.4-1.9%]; p = 0.52), or percentage of interstitial lung abnormalities (1.1% [CI90 -2.2-4.3%]; p = 0.58).
Image: PD
©2018 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.


![The ABCD2 score: Risk of stroke after Transient Ischemic Attack (TIA) [Classics Series]](https://www.2minutemedicine.com/wp-content/uploads/2013/05/web-cover-classics-with-logo-medicine-BW-small-jpg-350x250.jpg)