Teprotumumab may significantly improve outcomes in patients with active thyroid eye disease
1. In this phase 3 trial, patients with thyroid eye disease who received teprotumumab were far more likely to demonstrate a proptosis response compared to those who received placebo. On average, those who received teprotumumab also experienced a greater magnitude of response.
2. Patients in the teprotumumab group also experienced superior outcomes with regard to diplopia response, Clinical Activity Score and quality of life compared to patients in the placebo group.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Graves’ ophthalmopathy, also known as thyroid eye disease, is an autoimmune condition with numerous manifestations including eyelid retraction, proptosis, diplopia, and optic nerve compression. While the disease mechanisms are not completely understood, insulin-like growth factor I receptor (IGF-IR) is overexpressed in thyroid eye disease and is known to contribute to hyaluronan accumulation and cytokine expression, leading to edema of extraocular tissue. The monoclonal antibody teprotumumab has been shown to be an effective inhibitor in a phase 2 trial, and this phase 3 OPTIC trial aimed to confirm these findings, specifically focusing on reductions in proptosis as the main outcome of interest. Compared to those who received placebo, those who received teprotumumab had superior proptosis response and diplopia response as well as greater reductions in a 7-component Clinical Activity Score. Mild adverse events occurred in a higher percentage of patients in the treatment group, but only 1 serious adverse event related to the trial drug was observed. Of note, auditory issues arose in over 10% of the treatment group but all resolved within 21 days of the last dose. The primary limitation of this study was the lack of long-term follow-up, which precluded analysis of the durability of efficacy.
Click here to read the study in NEJM
Relevant Reading: Current and Future Treatments for Graves’ Disease and Graves’ Ophthalmopathy
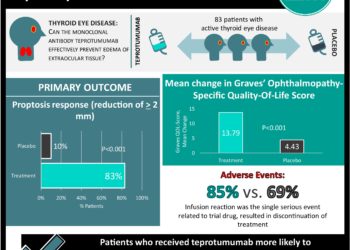
In-Depth [randomized controlled trial]: In this double-masked, placebo-controlled, multicenter trial involving patients with active thyroid eye disease, 83 patients were randomly assigned in a 1:1 ratio to receive either IV infusions of teprotumumab (10 mg/kg body weight for the first infusion and 20 mg/kg for subsequent infusions) or placebo once every 3 weeks for 21 weeks. Baseline characteristics including sex, age, race, duration of disease, proptosis measurement, and thryroid hormone level were similar between groups. The primary outcome of proptosis response was defined as a reduction of ≥2 mm and was observed in 34 patients (83%) in the treatment group versus 4 patients in the control group (P<0.001). All secondary outcomes, including overall response (78% of patients vs. 7%), Clinical Activity Score of 0 or 1 (59% vs. 21%), the mean change in proptosis (−2.82 mm vs. −0.54 mm), diplopia response (68% vs. 29%), and the mean change in Graves’ ophthalmopathy-specific quality-of-life overall score (13.79 points vs. 4.43 points), were strongly in favor of teprotumumab (P≤0.001 for all). Of note, 85% of patients in the teprotumumab group experienced any adverse event versus 69% in the placebo group. An infusion reaction was the single serious event related to the trial drug and resulted in discontinuation of treatment.
Image: PD
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.