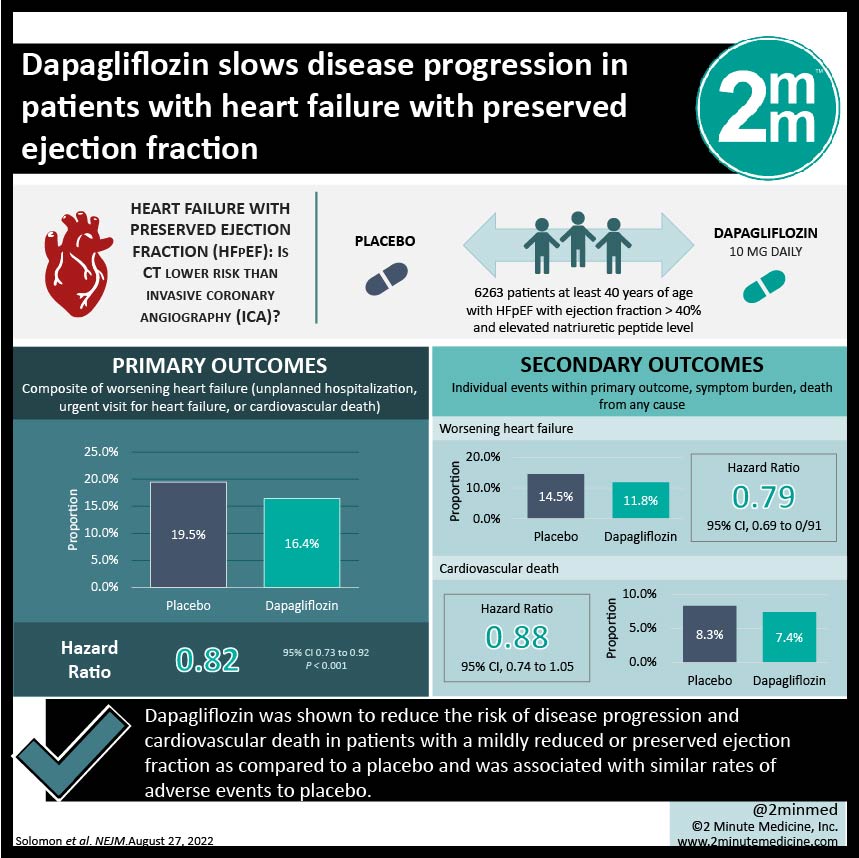
#VisualAbstract: Dapagliflozin slows disease progression in patients with heart failure with preserved ejection fraction
1. Dapagliflozin was shown to reduce the risk of disease progression and cardiovascular death in patients with a mildly reduced or preserved ejection fraction as compared to a placebo.
2. Dapagliflozin was associated with similar rates of adverse events to the placebo control.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Sodium-glucose cotransporter 2 (SGLT2) inhibitors, such as dapagliflozin, are well-established therapies used to reduce the risk of hospitalization and cardiovascular death in patients with heart failure and a left ventricular ejection fraction (LVEF) of 40% or less. However, it is unclear if SGLT2 inhibitors are beneficial for heart failure patients with higher LVEF. The current study was a phase three multicenter trial that aimed to investigate the efficacy and safety of dapagliflozin in treating heart failure patients with LVEF great than 40%. The control group was given a placebo. Dapagliflozin recipients were at significantly lower risks of worsening heart failure and cardiovascular disease, along with reduced symptom burden, compared to placebo recipients. This benefit was observed regardless of patients’ diabetic status or LVEF. The incidence of adverse events, including death, was comparable between the dapagliflozin and placebo groups. Although, the specificity of the inclusion and exclusion criteria may impact the generalizability of the study results. The present study provided evidence to support the use of dapagliflozin in heart failure patients with high LVEF.
Click here to read the study in NEJM
In-Depth [randomized controlled trial]: The current study was a phase three multicenter, double-blind randomized controlled trial aimed at assessing the efficacy and safety of dapagliflozin in heart failure patients with LVEF greater than 40%. Patients were included if they were at least 40 years of age, had stabilized heart failure, had an LVEF higher than 40%, had evidence of structural heart disease, and had an elevated natriuretic peptide level, regardless of their type 2 diabetes mellitus status. Exclusion criteria included previous use of an SGLT2 inhibitor, impaired kidney function, history of myocardial infarction, coronary revascularization, and certain causes of heart failure. In total, 6233 patients were randomized 1:1 to receive dapagliflozin 10mg once daily or a placebo. The primary outcome was a composite of worsening heart failure, which was defined as either an unplanned hospitalization, an urgent visit for heart failure, or cardiovascular death. Secondary outcomes included the individual events within the primary outcome, symptom burden, and death from any cause. Over a median follow-up of 2.3 years, the primary outcome occurred in 513 of 3,131 dapagliflozin recipients (16.4%) and in 610 of 3,132 placebo recipients (19.5%) (Hazard Ratio [HR], 0.82; 95% Confidence Interval [CI], 0.73 to 0.92; p<0.001). Worsening heart failure occurred in 11.8% of dapagliflozin recipients and 14.5% of placebo recipients (HR, 0.79; 95% CI. 0.69 to 0.91), while cardiovascular death occurred in 7.4% and 8.3% of patients, respectively (HR, 0.88; 95% CI, 0.74 to 1.05). Notably, the impact of dapagliflozin on primary outcome remained consistent regardless of whether patients had type 2 diabetes mellitus. The incidence of adverse events was comparable between the dapagliflozin and placebo groups. Although its generalizability may be impacted by the specific inclusion and exclusion criteria, the study provided robust evidence to support the use of dapagliflozin in treating heart failure patients with preserved LVEF.
©2022 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.