Benefits of high-intensity surveillance following colorectal adenoma removal likely outweigh costs
1. In this microsimulation model of U.S. patients aged 50-79, high-intensity surveillance following colorectal adenoma provided clinically significant benefits over low-intensity surveillance at acceptable cost.
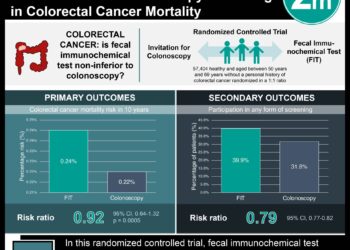
2. In nearly all evaluated alternative scenarios, the absolute cost-effectiveness of high-intensity surveillance remained under $100,000 per quality-adjusted life-year (QALY) gained. Compared with low-intensity surveillance, high-intensity surveillance cost less than $30,000 per QALY gained.
Evidence Level: 2 (Good)
Study Rundown: Colorectal cancer (CRC) is the 2nd leading cause of cancer death in the United States despite robust evidence pointing toward routine screening as an effective means of decreasing the risk of death through early detection and removal of precancerous adenomas. Current guidelines recommend surveillance after 5-10 years for patients with low-risk adenomas (LRA) and after 3 years for patients with high-risk adenomas (HRA). However, this frequency constitutes a significant strain on resources, prompting discussion regarding cost-effectiveness especially in light of improvements in colonoscopy performance. Previous observational studies have generally neglected the impact of surveillance intensity on risk of cancer death following adenoma resection, and relevant clinical trial results are not expected for several years. Findings from this microsimulation model supported existing guidelines for surveillance frequency for patients with HRAs and served to counterbalance previous modeling studies advocating against surveillance in patients with LRAs. Furthermore, this model suggested that compared to low-intensity surveillance, high-intensity surveillance yields continually increasing returns in the long term at acceptable cost. The main limitation of this study arose as a result of the dearth of long-term randomized trials concerning adenoma recurrence and CRC incidence. Consequently, despite extensive external model validation, it was difficult to accurately predict the course of CRC incidence at an individual level. Additionally, patients with LRAs of atypical topography or histology were not distinguished from their lower-risk counterparts, increasing the group’s overall likelihood of recurrence/incidence and potentially skewing results in favor of high-intensity surveillance.
Click to read the study in Annals of Internal Medicine
Click to read an accompanying editorial in Annals of Internal Medicine
Relevant Reading: Use of Colonoscopy to Screen Asymptomatic Adults for Colorectal Cancer
In-Depth [prospective cohort]: This study utilized a predictive model of CRC representative of average risk U.S. patients aged 50-79 who had adenomas removed at screening with colonoscopy or fecal immunochemical testing. Data was drawn from the NCI Surveillance, Epidemiology, and End Results (SEER) program. In analysis, adenomas were classified as small (<10 mm in diameter) or large (≥10 mm in diameter). Patients with LRAs were defined as having no more than 2 small adenomas, while patients with HRAs were defined as having between 3 and 10 small adenomas or at least one large adenoma. Model detection rates were validated against numerous chemoprevention trials and prospective cohort studies, model incidence predictions were validated against the largest retrospective cohort study on CRC incidence to date, and model mortality predictions were validated against two of the largest, most-cited studies on mortality. Primary study outcomes included lifetime CRC incidence/mortality, quality-adjusted life-years (QALY) gained, and cost-effectiveness ratios. In sensitivity analyses, the vast majority of evaluated scenarios resulted in a cost-effectiveness ratio significantly lower than the threshold, defined as $100,000/QALY gained. The only scenarios to exceed this threshold were high-intensity surveillance of patients with LRAs removed at age 70 years ($103,900/QALY gained) and near-perfect colonoscopy quality ($455,600/QALY gained).
Image: PD
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.

![2MM: AI Roundup- AI Cancer Test, Smarter Hospitals, Faster Drug Discovery, and Mental Health Tech [May 2nd, 2025]](https://www.2minutemedicine.com/wp-content/uploads/2025/05/Untitled-design-350x250.png)