Nanoparticle delivery of aurora kinase inhibitor may improve tumor treatment [PreClinical]
1. Nanoparticle delivery of the aurora kinase inhibitor AZD2811 led to sustained drug retention in a rat model of colon adenocarcinoma.
2. As compared to treatment with the free, soluble form of AZD2811, nanoparticle administration significantly decreased tumor size and delayed the onset of tumor regrowth.
Evidence Rating Level: 2 (Good)
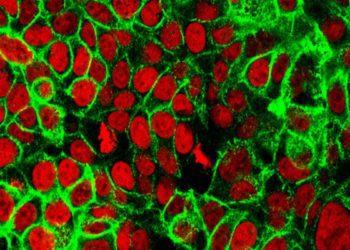
Study Rundown: Aurora kinase inhibitors (AKIs) are a class of pharmacological agents designed to target tumor cell proliferation. While effective in inducing tumor cell death, AKIs have not been successful in the clinic due to major off-target toxicity effects. In this study, the authors developed a nanoparticle formulation of the AKI AZD2811 using polymer materials in order to improve treatment efficacy while limiting cytotoxicity.
Initial studies identified optimized nanoparticle formulations that would increase drug load and decrease drug release rate. Two drug-nanoparticle compound formulations made with different solvents, Accurin B and Accurin E, were tested in a rat model of colon adenocarcinoma. As compared to treatment with the prodrug AZD1152 (AZD2811 in its free, water-soluble form), nanoparticle delivery of AZD2811 resulted in improved drug retention, extending the detection of AZD2811 in tumors from 24 hours to six days after final treatment administration. Nanoparticle treatment also prolonged the antimitotic effect of the AKI. Additionally, Accurin B and Accurin E prevented tumor growth almost twice as well AZD1152. Immunohistology of bone marrow samples showed that nanoparticle treatment reduced the severity of short-term off-target toxicity caused by AZD1152. Blood tests confirmed that Accurin E caused little impact on white blood cell counts and myeloid cell numbers. Finally, the researchers additionally tested Accurin E in two mouse models of diffuse large B cell lymphoma (DLBCL) and demonstrated that nanoparticle treatment successfully delayed tumor regrowth.
Overall, these preliminary studies demonstrated the feasibility of using nanoparticle delivery of AKIs to treat tumor growth while minimizing off-target toxicity. Because the materials used in these nanoparticles are FDA-approved, progression of this treatment to human clinical trials is promising. However, more preclinical testing to establish improved survival rates, for example, is necessary.
Click to read the study in Science Translational Medicine
Click to read an accompanying editorial in Science Translational Medicine
Relevant Reading: MK-0457, a novel kinase inhibitor, is active in patients with chronic myeloid leukemia or acute lymphocytic leukemia with the T315I BCR-ABL mutation
In-Depth [animal study]: In nude athymic rats grafted with human colon adenocarcinoma tumors, 25mg/kg of AZD1152, Accurin B, or Accurin E was administered intravenously. AZD2811 was present in resected tumors collected 6 days after final nanoparticle administration, while drug was not detected in samples from animals treated with the prodrug even 24 hours after treatment.
By measuring levels of phosphorylated histone H3 (pHH3), a marker of cell division, the authors found that AZD1152 treatment led to decreased cell proliferation after the first 6 hours post-treatment. However, pHH3 levels returned to levels comparable to that of the placebo-treated group (treated with empty nanoparticles) within 24 hours. In contrast, Accurin B and Accurin E sustained lowered pHH3 levels for up to 48 and 96 hours, respectively.
Tumor volume measurements 17 days after initial treatment determined that Accurin B and Accurin E treatment lead to decreased growth by 92% and 101%, respectively, as compared to AZD1152 treatment, which resulted in only a 58% volume reduction (n=10). Using immunohistochemistry, the cellularity of bone marrow samples was scored from 0 (no change) to 4 (severe). AZD1152 caused mild-to-moderate change (2-3) 5 days after treatment. In contrast, nanoparticle administration showed no-to-minimal change (0-1) with nanoparticle administration. Accurin E also decreased white blood cell numbers to a range comparable to those of placebo nanoparticles (n=2 to 5).
Finally, several additional xenograft mouse models were used to validate the findings. A colon adenocarcinoma cell line and two DLBCL cell lines were subcutaneously injected into mice to establish tumors. In all three models, Accurin E was able to significantly delay the regrowth of tumors as compared to placebo treatment and AZD1152 administration (n=10 to 12).
Image: PD
©2016 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.



![Nanoparticle delivery of aurora kinase inhibitor may improve tumor treatment [PreClinical]](https://www.2minutemedicine.com/wp-content/uploads/2016/02/20541_lores-75x75.jpg)
![Drug reformulation may improve treatment of Niemann-Pick type C disease [PreClinical]](https://www.2minutemedicine.com/wp-content/uploads/2016/02/Niemann_pick_cell_in_spleen-75x75.jpg)
