Upadacitinib monotherapy is effective for patients with moderate-to-severe atopic dermatitis
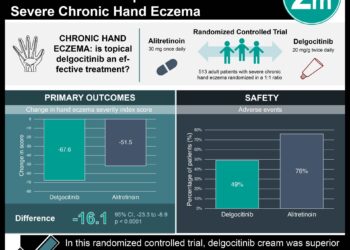
1. The proportion of patients with EASI ≥75% was significantly greater in the upadacitinib 30 mg and 15 mg groups than the placebo group.
2. Both upadacitinib doses were well tolerated, with the most common adverse events being acne and upper respiratory tract infection.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Upadacitinib is an oral Janus kinase (JAK) inhibitor that has been approved for the treatment of moderate to severe rheumatoid arthritis. However, its use for treatment of other immune-mediated diseases is currently under investigation. This randomized controlled trial aimed to assess the efficacy and safety of upadacitinib compared with placebo for the treatment of moderate-to-severe atopic dermatitis in adolescents and adults. Coprimary efficacy endpoints were proportion of patients with ≥ 75% improvement in Eczema Area and Severity Index (EASI) score from baseline and patients with validated Investigator’s Global Assessment for Atopic Dermatitis (vIGA-AD) response at week 16. Secondary endpoints included proportion of patients with Worst Pruritus Numerical Rating Scale (WP-NRS) ≥ 4, and those with EASI ≥ 90%. According to study results, monotherapy with upadacitinib had a positive benefit-risk profile in adolescents and adults with moderate-to-severe atopic dermatitis. More specifically, both coprimary endpoints were significantly improved with upadacitinib monotherapy while having a comparable safety profile to placebo. A major strength of this study was the inclusion of separate randomized controlled trials (RCTs) with different age groups (adolescents and adults) given the disease burden of atopic dermatitis in each cohort. These findings indicate the potential for upadacitinib to improve treatment in patients with moderate-to-severe atopic dermatitis.
Click to read the study in The Lancet
Relevant Reading: Trial of Upadacitinib and Adalimumab for Psoriatic Arthritis
In-depth [randomized controlled trial]: Between Aug 13, 2018, and Dec 23, 2019, 1093 patients were assessed for eligibility in the Measure Up 1 (MU1) study, and 1143 patients were assessed in the Measure Up 2 (MU2) study (between July 27, 2019, and Jan 17, 2020). Included patients were adolescents (aged 12-17 years) and adults (aged 18-75 years) with moderate-to-severe atopic dermatitis (defined by ≥ 10% of body surface area affected by atopic dermatitis, EASI ≥ 16, vIGA-AD ≥ 3, and WP-NRS ≥ 4). Patients were randomly assigned to receive oral upadacitinib 15 mg, 30 mg, or placebo once daily for 16 weeks. Altogether, 847 patients in MU1 and 836 patients in MU2 were included in the efficacy and safety analyses.
In MU1, the mean participant age was 34.0 years (standard deviation [SD] 15.7) and 54% were male; whereas in MU2, the mean age was 33.6 years (SD 15.5) and 56% were male. At week 16, coprimary endpoints were met for both trials. The proportion of patients with EASI ≥ 75% for MU1 was significantly greater in the upadacitinib 30 mg (80%) and 15 mg (70%) groups than the placebo (16%) group. This was also the case for patients in MU2, with 73% of patients in upadacitinib 30 mg and 60% in upadacitinib 15 mg having EASI ≥ 75%, compared to 13% in the placebo group. The proportion of patients who achieved a vIGA-AD response at week 16 was also significantly higher in the upadacitinib 30 mg (62%) and 15 mg (48%) groups than the placebo (8%) group (MU1). In MU2, vIGA-AD responses were highest in upadacitinib 30 mg (52%) followed by 15 mg (39%) and placebo (5%). Both upadacitinib doses were well-tolerated with the most frequently reported adverse event being acne (17% upadacitinib 30 mg MU1 vs. 15% upadacitinib 30 mg MU2).
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.