Eculizumab treats atypical hemolytic-uremic syndrome
Image: PD
1. Treatment with the anti-C5 monoclonal antibody eculizumab significantly improved hematologic markers and GFR in patients with atypical hemolytic uremic syndrome.
2. The improvements in hematologic markers and GFR were sustained beyond the duration of therapy.
Evidence Rating Level: 2 (Good)
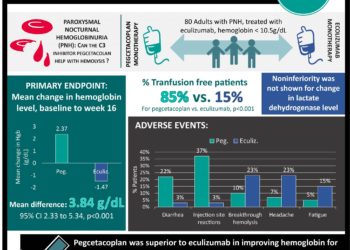
Study Rundown: Atypical hemolytic-uremic syndrome (AHUS) is a genetic disease caused by a defect in the regulation of complement. Disregulated complement activates platelets causing systemic microvascular disease, called thrombotic microangiopathy (TMA) resulting in damage to multiple organs including the kidney. The authors of this paper performed two small phase 2 trials examining the efficacy and safety of Eculizumab, a monoclonal antibody that targets complement protein C5. This open-label study enrolled patients with a clinical diagnosis of AHUS, and compared baseline measurements of platelets, GFR, and other markers with values after 26 weeks of therapy and 62-64 weeks of follow up. Eculizumab therapy appeared to significantly inhibit complement-mediated TMA, and also appeared to improve renal function beyond the course of the therapy. While adverse events were reported, none interfered with treatment, and no severe infection-related events occurred. These were small single-group trials that did not differentiate between patient etiologies of AHUS. However, given the small number of patients with the disease, and structured comparison of measurable markers to baseline, the trial appears reasonably performed to demonstrate efficacy and safety of eculizumab as a treatment for AHUS.
Click to read the study, published today in NEJM
Relevant Reading: Atypical Hemolytic-Uremic Syndrome
In-Depth [prospective phase 2 trials]: These studies investigated the efficacy and safety of eculizumab when used to treat AHUS. The studies analyzed data from a total of 32 patients with the clinical diagnosis of AHUS. Trial 1 focused on patients with progressive thrombotic microangiopathy (evidenced by a decreasing platelet count despite standard therapy of plasma exchange or infusion). 13 patients completed a 26 week course of treatment with eculizumab and completed long-term follow up. The primary end points for trial 1 were inhibition of TMA (indicated by increased platelet count) and normalization of hematologic values including platelets and LDH. After a median follow up of 64 weeks, 88% of patients in trial 1 had normal platelet counts and normal LDH. Trial 2 focused on patients whose platelet counts were stable on standard therapy, but still had impaired renal function and evidence of hemolysis.19 patients were treated for 26 weeks and were followed up for a median duration of 62 weeks. The primary end points for trial 2 were TMA event-free status for at least 12 weeks, and normalization of hematologic values. After long term follow up, 85% of patients had TMA event-free status, and 90% had normal hematologic values. In both trials, patients showed an improvement in GFR and decease in proteinuria from baseline. In trial 1, 4 out of 5 patients discontinued dialysis. All patients in trial 1 had at least one serious adverse event. 50% of patients in trial 2 had serious events. All events possibly related to eculizumab resolved without interruption of treatment and no new events were reported after completion of the therapy.
By Akira Shishido, M.D. and Mitalee Patil
More from this author: Linezolid treats Chronic Extensively Drug-Resistant TB but has a high incidence of adverse effects, Intermittent and continuous antiretroviral therapies are equivalent in the treatment of acute HIV-1 infection, Peginesatide is noninferior to epoetin for anemia in hemodialysis patients [EMERALD Trials], Antibiotics improve outcomes in severe acute malnutrition, Carbapenem resistance in Enterobacteriaceae nearly quadrupled in the last decade, new study finds.
© 2013 2minutemedicine.com. All rights reserved. No works may be reproduced without written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT. Content is produced in accordance with fair use copyrights solely and strictly for the purpose of teaching, news and criticism. No benefit, monetary or otherwise, is realized by any participants or the owner of this domain.