Ponesimod may be superior to teriflunomide for relapsing multiple scleorsis
1. Ponesimod was superior to teriflunomide for relapsing multiple sclerosis, in terms of reducing relapse rates, unique active lesions on MRI, and fatigue symptoms.
Evidence Rating Level: 1 (Excellent)
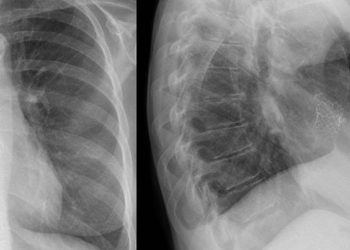
Multiple sclerosis (MS) is an autoimmune, chronic disease where demyelination and loss of axons manifest in relapses and recurrent attacks, leading to impaired neurological function. More investigations into MS treatments that are safe, effective, and convenient are still needed. The current double-blind, randomized controlled trial examined the efficacy, safety, and tolerability of two oral treatments that have been approved for recurrent MS: Ponesimod, a selective sphingosine-1-phosphate receptor modulator, and teriflunomide, a pyrimidine synthesis inhibitor. The study population consisted of 1133 patients across 28 countries, 64.9% female with a median (range) age of 37.0 (18-55) years. In terms of relapses, there were 242 relapses in the ponesimod group and 344 for teriflunomide, with ponesimod reducing the annualized relapse rate (ARR) by 30.5% (mean ARR 0.202 vs 0.290; rate ratio 0.695, 99% CLs 0.536-0.902; p < 0.001). Furthermore, a symptom score assessing fatigue in MS patients (the FSIQ-RMS) showed lower levels of fatigue at week 108 for ponesimod (mean difference -3.57, 95% CLs -5.83 to -1.32, p = 0.002). Ponesimod also reduced the mean number of combined unique active lesions per year by 56%, as assessed on MRI (1.405 vs 3.164; rate ratio 0.444, 95% CLs 0.346-0.542; p < 0.001). However, the difference in 12-week and 24-week confirmed disability accumulation risk estimates were found to be not significantly different. Overall, this study demonstrated the superiority of ponesimod to teriflunomide in terms of efficacy in reducing relapses and tolerability of symptoms.
Click to read the study in JAMA Neurology
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.