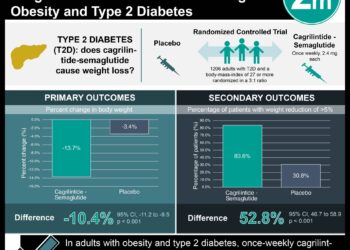
The ADVANCE trial: Intensive glycemic control reduces risk of nephropathy [Classics Series]
1. Intensive glycemic control was associated with significantly lower rates of new or worsened nephropathy when compared to conventional glycemic control.
2. There was no significant reduction in major macrovascular events associated with intensive blood sugar control.
Original Date of Publication: June 12, 2008
Study Rundown: Glycated haemoglobin (HbA1c) levels are frequently used as a marker of glycemic control in diabetic patients. Previous studies, such as the ACCORD and UKPDS trials, have demonstrated that tighter glycemic control reduces the risk of microvascular complications (i.e., nephropathy, retinopathy, neuropathy). The ACCORD trial, however, also noted that there was significantly higher risk of mortality in patients who underwent tight glycemic control (i.e., target HbA1c <6.0%). Moreover, there is no strong evidence demonstrating that better glycemic control significantly improves rates of macrovascular complications (i.e., myocardial infarction, stroke).
The Action in Diabetes and Vascular Disease: Preterax and Diamicron Modified Release Controlled Evaluation (ADVANCE) trial sought to assess the effects of intensive glycemic control (i.e., target HbA1c ≤6.5%) on vascular outcomes. The findings demonstrate that patients in the intensive group had significantly lower risk of new/worsening nephropathy (HR 0.79; 95%CI 0.66-0.93) and new-onset microalbuminuria (HR 0.91; 95%CI 0.85-0.98) when compared with standard therapy. There were no significant differences between the two groups in rates of macrovascular complications or all-cause mortality. Importantly, the risk of severe hypoglycaemia was significantly higher in patients undergoing intensive therapy (HR 1.86; 95%CI 1.42-2.40).
Click to read the study in NEJM
In-Depth [randomized controlled study]: The study included 11,140 participants from 215 centres in 20 countries. Patients were eligible if they were ≥55 years of age, were diagnosed with type 2 diabetes mellitus at ≥30 years of age, and had a history of micro- or macrovascular disease. Exclusion criteria included a definite indication for or contraindication to any of the study drugs, or a definite indication for long-term insulin therapy at study entry. Included patients were randomized to either intensive glucose control (i.e., target HbA1c ≤6.5%) or standard glucose control (i.e., based on local guidelines). The intensive control group received gliclazide and other adjuvants to achieve target HbA1c, while the control group received treatment as per local guidelines. The primary outcome was a composite of macrovascular (i.e., myocardial infarction, stroke, or death from cardiovascular event) and microvascular events (nephropathy or retinopathy).
Median follow-up time was 5 years. At the end of the follow-up period, the mean HbA1c levels were 6.5% and 7.3% in the intensive and standard groups, respectively. There were no significant differences between the two groups with regards to the incidence of major macrovascular events (HR 0.94; 95%CI 0.84-1.06) and death from any cause (HR 0.93; 95%CI 0.83-1.06). The intensive group had a significantly lower rate of major microvascular events (HR 0.86; 95%CI 0.77-0.97), which was driven by a significantly lower risk of new or worsening nephropathy (HR 0.79; 95%CI 0.66-0.93) and new-onset microalbuminuria (HR 0.91; 95%CI 0.85-0.98). There was no significant difference between the two groups in terms of the risk of new or worsening retinopathy. The risk of severe hypoglycaemia, however, was significantly higher in the intensive group (HR 1.86; 95%CI 1.42-2.40).
Image: PD
©2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.




![[Physician Comment] Suture closure superior to staples after cesarean delivery](https://www.2minutemedicine.com/wp-content/uploads/2013/01/xAtraumatisches_Nahtmaterial_17-e1356930998526.jpg.pagespeed.ic_.9-GxOSos0V-75x75.jpg)
![[Physician Comment] Pneumocystis linked to Sudden Unexpected Infant Deaths](https://www.2minutemedicine.com/wp-content/uploads/2013/01/xPneumocystis-e1357225414200.jpg.pagespeed.ic_.pmvU9z1gtu-75x75.jpg)