2 Minute Medicine Rewind January 3, 2021
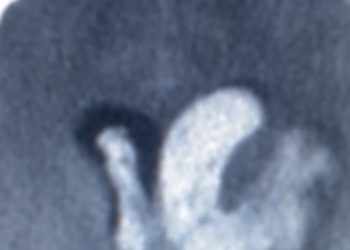
1. Carboplatin, given through intracerebral, convection-enhanced delivery, may be a viable adjunct in recurrent high-grade glioma treatment.
Evidence Rating Level: 1 (Excellent)
Patients with glioblastomas (GBM), which will invariably recur despite maximal therapy, have a poor prognosis, with a median overall survival of 15 months. The current treatment for newly diagnosed GBM includes surgical resection, concurrent radiation, and temozolomide with subsequent adjuvant temozolomide. Carboplatin is highly effective in treating other malignancies. However, while gliomas have been shown to be sensitive to this anti-cancer drug, using carboplatin has been limited by its systemic toxicity and its inability to penetrate the blood-brain barrier at effective concentrations. In this open-label, nonrandomized dose-escalation trial, 10 patients with recurrent WHO grade III or IV gliomas, who had previously undergone surgery and chemoradiation, were divided into cohorts of 3 patients each. They were treated with escalating doses of carboplatin at 1ug, 2ug, and 4ug in 54mL, delivered intracerebrally via convection enhanced delivery (CED) over 72 hours. No treatment-related deaths occurred, and no dose-limiting toxicities were observed. Neither were reported adverse events (total of 9) considered treatment-related, aside from one patient which experienced an isolated grade 2 generalized tonic-clonic seizure; this incident was resolved after halting carboplatin infusion (1.3ug dose) and the patient recovered to baseline medical status after infusion was halted within 24 hours. Progression-free survival at 6 months post-infusion was 20% (median 2.1 months) and 12-month overall survival was 40% (median 9.6 months). It is of note however, that the study’s inclusion and exclusion criteria led to a predominantly younger cohort (mean age= 46.8years) with high baseline KPS (Karnofsky performance score), may have contributed to the low incidence of adverse events and the trend towards more favourable survival. While this study shows intracerebral CED of carboplatin of up to 4ug is a safe and feasible adjunct to re-resection of recurrent GBMs, further research is needed with a larger sample size to determine maximum tolerated dose and the generalizability of these results.
Perioperative changes of response to antiplatelet medication in vascular surgery patients
1. The incidence of having reduced antiplatelet activity to aspirin (ALR) increased after vascular procedures.
Evidence Rating Level: 1 (Excellent)
Though clinical guidelines often recommend antiplatelet therapy in secondary and tertiary prevention of thromboembolic events, studies investigating their efficacy have shown variable results. Moreover, while vascular surgery patients are at high risk of these events, few studies have evaluated the prevalence of reduced antiplatelet activity of aspirin (ALR) or clopidogrel (CLR) in this population, and any changes in ALR or CLR which may occur after their procedures. In this prospective, observational, single-centre study, 176 patients taking either aspirin 100 mg daily, aspirin 300 mg daily, or clopidogrel 75 mg daily with symptomatic PAD (peripheral arterial disease) or internal carotid stenosis were included. They had the activity of their antiplatelet medication measured by impedance aggregometry (using Multiplate®) before and after their vascular procedure. Prior to vascular treatment, prevalence rates of ALR and CLR were 13.1% and 32% respectively. Post-operatively, the overall ALR increased significantly by approximately two-fold (27.5%, p=0.0006), while no significant change in CLR was detected. Due to low numbers of cases in the other groups, risk factor analysis was only performed in patients taking aspirin 100 mg daily. In this cohort, concomitant insulin medication (p=0.0006) and elevated C-reactive protein (p=0.0021) were found to be potential risk factors for having a low response rate to antiplatelet therapy. Overall, this study showed ALR prevalence in vascular surgery patients to be at the lower end of values reported in literature; however, the comparability of these results are limited as there is no preferred diagnostic test in detecting low antiplatelet response. How these reduced antiplatelet response rates may or may not affect clinical outcomes will require gathering long-term data and whether other potential risk factors may contribute to ALR and CLR will require studying larger vascular surgery populations.
1. Stopping renin-angiotensin system inhibition in patients with advanced chronic kidney disease was associated with higher risk of mortality, major cardiovascular events, and lower risk of kidney replacement therapy.
Evidence Rating Level: 2 (Good)
Studies have shown that renin-angiotensin system inhibitors (RASi) are effective in delaying chronic kidney disease (CKD) progression. Angiotensin-converting enzyme inhibitors (ACEis) and angiotensin II receptor blockers (ARBs) are integral to treating proteinuric CKD. However, it is unknown whether RASi are safe and effective in patients with advanced CKD, or with eGFR <30mL/min/1.73m2. While continuing RASi could accelerate the need for kidney replacement therapy (KRT), stopping RASi may impact its cardioprotective and mortality benefits. In this observational study, 10 254 patients from the Swedish Renal Registry, taking RASi for more than 80% of the 2 years before January 1, 2007 and with new-onset CKD G3-5, were included. Researchers emulated a target trial with a 5 year follow-up, using techniques such as cloning, censoring, and weighting, to compare the effects of stopping RASi within 6 months after eGFR dropped below 30mL/min/1.73m2 compared to continuing RASi. From this cohort (median age 72 years, median eGFR 23mL/min/1.73m2), 15% stopped RASi therapy within 6 months. RASi discontinuation was associated with an increase in both absolute 5-year risk of death (54.5% vs 40.9%, ARD 13.6) and in major adverse cardiovascular events (59.5% vs 47.6%, ARD 11.9) over those who continued RASi treatment. However, stopping RASi therapy was also associated with a lower risk of KRT (27.9% vs 36.1%, ARD -8.3). These results remained consistent regardless of whether patients stopped RASi at an eGFR higher or below 20mL/min/1.73m2, after adjusting and stratifying for albuminuria and potassium, and after modelling RAS inhibition as a time-dependent exposure on a marginal structural model. This study supports that the decision to stop RAS inhibition in patients with advanced CKD should be made after evaluating cardiovascular and KRT risk. Variables not captured in this trial include the reason for RASi discontinuation, such as patient frailty, and variation in practitioner behaviour: for example, the initiation of KRT itself is a treatment decision. Further study, especially randomized trials, should be conducted in order to support these observational findings.
1. Positive fecal immunochemical test (FIT) results were associated with increased incidences of ischemic stroke, myocardial infarction, and all-cause mortality.
Evidence Rating Level: 2 (Good)
While fecal immunochemical tests (FIT) is a commonly used and effective screening tool for colorectal cancer (CRC), their clinical use in detecting other diseases is poorly known. As cardiovascular diseases and CRCs share risk factors such as old age, smoking, and physical inactivity, these conditions may share an underlying pathogenesis (i.e. systemic inflammation). Therefore, FIT results may share some insight into cardiovascular morbidity and all-cause mortality. In this nationwide population-based cohort study, 6 277 446 participants (over 50 years old) with CRC screening records from 2009-2012 in the South Korean National Health Insurance database were included. Patients with a history of cardiovascular disease and/or CRC were excluded. Over a median follow-up of 6.79 years, incidence rates of both ischemic stroke (4.88 vs 3.90 per 1000 patient years) and myocardial infarction (2.99 vs 2.44 per 1000 patient years) were higher in the FIT-positive over the FIT-negative group. This increased risk of ischemic stroke (aHR 1.25, 95%CI 1.23-1.27) and MI (aHR 1.09, 95% CI 1.19-1.25) remained significant after adjusting for demographic, social history, and comorbidity factors. Additionally, FIT-positive results were associated with increased all-cause mortality (aHR 1.15, 95% CI 1.07-1.23). Furthermore, these associated risks remained positive regardless of CRC and/or anemia status. By demonstrating an association between FIT positivity and increased stroke, MI, and all-cause mortality, these findings support a role for FIT beyond CRC screening. Nonetheless, randomized controlled trials would be needed to establish any causal relationships; future studies should also evaluate antithrombotic use, a common cause of false positive FIT results, and be capable of distinguishing between first-time and recurrent cardiovascular events.
Treatment with cardiovascular medications: Prognosis in patients with myocardial injury
1. Although infrequently prescribed, treatment with cardiovascular medications in patients with type 2 myocardial infarction (T2MI) and acute or chronic myocardial injury may lower mortality risk.
Evidence Rating Level: 2 (Good)
While patients with non-ischemic myocardial injury and type 2 myocardial infarction (T2MI) are associated with poorer outcomes and higher mortality, compared to patients with type 1 myocardial infarction (T1MI), there is ongoing debate regarding their treatment. Specifically, whether cardiovascular medications used in T1MI treatment would improve clinical outcomes in patients with other types of myocardial injury remains unknown. In this observational cohort study, records from 3 893 patients (mean age 73 years), taken from the Swedish National Patient Register, presenting with chest pain at the Karolinska University Hospital in Stockholm and with a discharge diagnosis of MI, were evaluated. Participants were divided into cohorts with T2MI, non-ischemic acute myocardial injury, and T1MI. They were then categorized according to the number of cardiovascular medications, calculated from all prescriptions dispensed within 180 days of their emergency department visit: those taking 0-1, 2-3, and 4 medications. Patients with T2MI and chronic myocardial injury were infrequently prescribed cardiovascular medications. For instance, approximately 15% of patients with T2MI and chronic myocardial injury did not receive any type of studied medicine (including ACEi, ARB, platelet inhibitors, and B-Blockers), such as only 40% of patients in these cohorts being given statins, compared to 87% of patients with T1MI. With a mean follow-up of 3.1+ 1.5 years, 27% of patients died, with yearly mortality rates decreasing with increasing numbers of medications across all types of myocardial injury. For example, patients with T2MI treated with 2-3 and 4 medications were associated with 50% (aHR 0.50, NSD) and 56% (0.43, 95% CI 0.19-0.96) lower rates of mortality than those taking 0-1 medications. These findings support a more generous approach to prescribing cardiovascular medications in patients with T2MI and acute or chronic myocardial injury may reap mortality benefit. However, this study was limited by its observational methods, lack of additional patient history to eliminate residual confounders, and its small number of patients and events in certain analyses (i.e. only 8 deaths in the reference group). Future studies should clarify diagnoses by accessing coronary angiographies and stress tests, and explore the effect of prescribed doses of specific medications.
Image: PD
©2020 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.


![2 Minute Medicine: Pharma Roundup: Price Hikes, Breakthrough Approvals, Legal Showdowns, Biotech Expansion, and Europe’s Pricing Debate [May 12nd, 2025]](https://www.2minutemedicine.com/wp-content/uploads/2025/05/ChatGPT-Image-May-12-2025-at-10_22_23-AM-350x250.png)