Sotagliflozin increase days-alive-and-out-of-hospital in patients with diabetes and heart failure
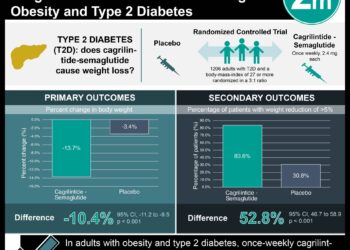
1. Sotagliflozin treatment was shown to increase the days-alive-and-out-of-hospital compared to placebo in patients with type 2 diabetes and heart failure.
2. Sotagliflozin treatment was shown to significantly decrease the rate of days dead compared to the placebo treatment.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Sodium-glucose cotransporter 2 (SGLT2) inhibitors have been shown to reduce the risk of hospitalization for heart failure among patients with diabetes. However, it remains unclear whether the use of these inhibitors is associated with changes in the duration of admission. As such, this study examined hospitalization rates along with the days-alive-and-out-of-hospital (DAOH) – a composite measure of mortality as well as both the frequency and duration of hospitalization. The study determined DAOH was increased among patients who received sotagliflozin, which was an effect primarily driven by a reduction in the rate of days dead. This study was limited by a lack of data regarding the primary reason for hospitalization, which precluded the investigation of disease-specific outcomes or potential adverse events. Nonetheless, these findings constitute additional evidence in support of SGLT inhibitors as a therapeutic option to reduce disease burden among patients with type 2 diabetes and worsening heart failure.
Click here to read the study in Annals of Internal Medicine
Relevant Reading: Sotagliflozin in Patients with Diabetes and Recent Worsening Heart Failure
In-Depth [randomized controlled trial]: The randomized control trial enrolled 1,222 patients between June 2018 and August 2020. Patients between the ages of 18 and 85 years, who had a previous diagnosis of type 2 diabetes, and had been hospitalized within two weeks of randomization for heart failure were included in the study. Patients who had not received intravenous diuretic therapy were excluded from the study. The patients were randomized in a 1:1 ratio to receive either sotagliflozin (200 mg, once daily) or placebo treatment, respectively. The primary endpoint was hospitalization for any reason and DAOH using prespecified Poisson regression models. Overall, the sotagliflozin group (16.3%) had fewer patients who were hospitalized more than once compared to the placebo group (22.1%). Despite this apparent benefit, the mean days in the hospital was shorter in the sotagliflozin group (1.9 per 100 days) compared to the placebo group (2.2 per 100 days) (rate ratio [RR], 0.86; 95% confidence interval [CI], 0.69 to 1.08; P = 0.21). However, the sotagliflozin group (6.3 per 100 days) showed a significant reduction in the rate of days dead compared to the control group (8.9 per 100 days) (RR, 0.71; 95% CI, 0.52 to 0.99; P = 0.041). Together, these differences yielded a DAOH rate that was higher in the sotagliflozin group (91.8 per 100 days) compared to the placebo group (88.9 per 100 days) (RR, 1.03; 95% CI, 1.00 to 1.06; P = 0.027). Altogether, sotagliflozin therapy was shown to increase the DOAH by lowering the rate of days dead compared to placebo treatment in patients with type 2 diabetes and worsening heart failure.
Image: PD
©2021 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc.