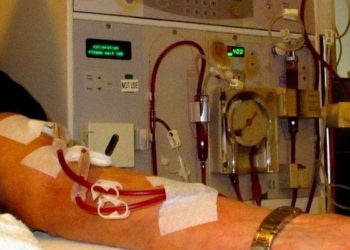
The PIVOTOL trial: high-dose iron reduces incidence of serious adverse events in hemodialysis patients
1. Patients undergoing maintenance hemodialysis experienced fewer serious adverse events if treated proactively with high-dose intravenous iron sucrose compared to patients reactively treated with low-dose iron sucrose.
2. Higher doses of erythropoiesis-stimulating agents were utilized more in the low-dose iron group compared to the high-dose group.
Evidence Rating Level: 1 (Excellent)
Study Rundown: Maintenance hemodialysis increases blood loss and reduces iron absorption, putting patients at risk for anemia. Intravenous iron administration is now utilized to counteract iron loss in hemodialysis patients, though what dose is optimal is presently unknown. The Proactive IV Iron Therapy in Haemodialysis Patients (PIVOTOL) trial sought to determine if proactive high-dose or reactive low-dose iron therapy would be safer in terms of minimizing serious adverse events, with results indicating patients in the high-dose group experienced fewer major adverse events. Additionally, erythropoiesis-stimulating agents were utilized less in the high-dose group, and infection rates were similar between groups.
This large randomized trial provides substantial evidence for supporting an optimal-dose treatment regimen involving important aspects of both safety and efficacy of iron treatment for hemodialysis patients. Its open-label design and lack of more dosing levels limits its results, though the long-term follow-up is a substantial strength.
Click to read the study in NEJM
Relevant Reading: Considerations and challenges in defining optimal iron utilization in hemodialysis
In-Depth [randomized controlled trial]: This open-label randomized controlled trial enrolled patients at multiple sites in the United Kingdom between 2013 and 2018. Eligible patients had end-stage kidney disease requiring maintenance hemodialysis which was initiated no more than 12 months prior to randomization and also had low ferritin and transferrin saturation levels. Patients were randomized to either proactive high-dose iron sucrose therapy (n=1093) or a reactive low-dose iron sucrose group (n=1048). Ferritin and transferrin saturation values were measured, which guided the iron doses received in both groups. Patients were also given erythropoiesis-stimulating agents as clinically directed to maintain a hemoglobin level within a prespecified target range. The primary endpoint of the trial, a composite outcome of myocardial infarction, stroke, hospitalization due to heart failure, and death, occurred in 29.3% of high-dose and 32.3% of low-dose patients (hazard ratio [HR], 0.85; 95% confidence interval [CI], 0.73 to 1.00; P<0.001 for noninferiority; P=0.04 for superiority) after a median follow-up of 2.1 years. With subgroup analysis, high-dose iron therapy was beneficial for all prespecified groups. Rates of hospitalization or infection were both similar between both groups. The median monthly dose of erythropoiesis-stimulating agent was significantly lower in the high-dose group (median difference, −7539 IU; 95% CI, −9485 to −5582). Serious adverse events occurred in 64.9% and 64.0% of patients in the high- and low-dose groups, respectively.
Image: PD
©2019 2 Minute Medicine, Inc. All rights reserved. No works may be reproduced without expressed written consent from 2 Minute Medicine, Inc. Inquire about licensing here. No article should be construed as medical advice and is not intended as such by the authors or by 2 Minute Medicine, Inc