The RENAAL Trial: Losartan in diabetic nephropathy [Classics Series]
Image: PD
1. In patients with type II diabetes and nephropathy, losartan at a dose of 50-100 mg daily significantly reduced the risk of developing end-stage renal disease compared to placebo.
2. While losartan also significantly reduced the degree of proteinuria in these patients, it did not significantly reduce mortality when compared with placebo.
Original Date of Publication: September 20, 2001
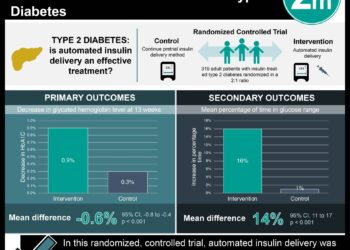
Study Rundown: Diabetic nephropathy is a leading cause of end-stage renal disease (ESRD). Previous studies had shown that blockade of the renin-angiotensin system slowed the progression of renal disease in patients with type I diabetes. The RENAAL (Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan) study was the one of the first to assess the effect of disrupting the renin-angiotensin system in patients with type II diabetes. The study demonstrated that in type II diabetic patients already receiving conventional anti-hypertensive therapy, the use of the angiotensin-II-receptor antagonist losartan significantly decreased the risk of ESRD (19.6% vs. 25.5%, 28% risk reduction, p=0.002). Losartan also significantly decreased the degree of proteinuria (p<0.001). There was no significant difference between the groups in mortality rates. One limitation of this study was the high rate at which patients discontinued the study drug. About 53.5% of patients in the placebo group and 46.5% of patients in the losartan group stopped taking their study medication early.
In summary, the findings of this study support the use of losartan in delaying the progression of renal disease in patients with type 2 diabetes and nephropathy.
Click to read the study in NEJM
In-Depth [randomized controlled trial]: Originally published in 2001 in NEJM, this randomized, controlled trial enrolled 1,513 patients from 250 centers in 28 countries. Eligible patients were between 31-70 years of age with diagnoses of type 2 diabetes and nephropathy (i.e., urinary protein ≥0.5 g/24 hours and serum creatinine between 115-254 μmol/L). Patients who had type 1 diabetes, non-diabetic renal disease, or a history of heart failure were excluded. Moreover, patients who had recent myocardial infarction (MI), percutaneous coronary intervention (PCI), coronary artery bypass grafting (CABG), or cerebrovascular event were excluded. Patients received conventional anti-hypertensive therapy as needed, in addition to either losartan or placebo. Permitted anti-hypertensive therapy included calcium-channel blockers, diuretics, alpha-blockers, and beta-blockers, but not angiotensin-converting-enzyme inhibitors (ACE-Is) or angiotensin-II-receptor antagonists other than losartan. Treatment was administered for a mean of 3.4 years. The primary outcome measure was time to the composite endpoint comprised of ESRD (i.e., need for dialysis or renal transplantation), doubling of serum creatinine level, and death. The secondary endpoints were morbidity and mortality from cardiovascular causes, progression of renal disease, and changes in the degree of proteinuria.
The daily dose of losartan ranged from 50-100 mg daily with 71% of patients receiving 100 mg. Losartan treatment significantly reduced the incidence of the primary endpoint when compared to placebo (43.5% vs. 47.1%, 16% risk reduction, p=0.02). This difference was driven by significant reductions in the risk of doubling serum creatinine (21.6% vs. 26.0%, 25% risk reduction, p=0.006) and the risk of end-stage renal disease (19.6% vs. 25.5%, 28% risk reduction, p=0.002) in the losartan group. There was no significant difference between the two groups in mortality (21.0% vs. 20.3%, p=0.88). Moreover, there was no difference between the groups in the secondary endpoint of morbidity and mortality from cardiovascular causes. Patients in the losartan group, however, did experience significant reductions in the amount of proteinuria when compared with those receiving placebo (p<0.001).
© 2012-2014 2minutemedicine.com. All rights reserved. No works may be reproduced without expressed written consent from 2minutemedicine.com. Disclaimer: We present factual information directly from peer reviewed medical journals. No post should be construed as medical advice and is not intended as such by the authors, editors, staff or by 2minutemedicine.com. PLEASE SEE A HEALTHCARE PROVIDER IN YOUR AREA IF YOU SEEK MEDICAL ADVICE OF ANY SORT.